a-Homonojirimycin
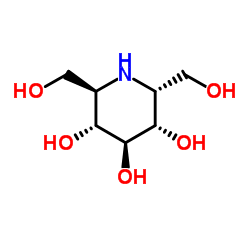
a-Homonojirimycin structure
|
Common Name | a-Homonojirimycin | ||
|---|---|---|---|---|
| CAS Number | 119557-99-2 | Molecular Weight | 193.198 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 432.0±45.0 °C at 760 mmHg | |
| Molecular Formula | C7H15NO5 | Melting Point | 207ºC | |
| MSDS | USA | Flash Point | 224.7±19.3 °C | |
Use of a-Homonojirimycinα-Homonojirimycin is a potent α-glucosidase inhibitor[1]. |
| Name | (2R,3S,5R,6R)-2,6-bis(hydroxymethyl)piperidine-3,4,5-triol |
|---|---|
| Synonym | More Synonyms |
| Description | α-Homonojirimycin is a potent α-glucosidase inhibitor[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 432.0±45.0 °C at 760 mmHg |
| Melting Point | 207ºC |
| Molecular Formula | C7H15NO5 |
| Molecular Weight | 193.198 |
| Flash Point | 224.7±19.3 °C |
| Exact Mass | 193.095016 |
| PSA | 113.18000 |
| LogP | -2.62 |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.583 |
| Storage condition | -20°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
~69% 
a-Homonojirimycin CAS#:119557-99-2 |
| Literature: Aoyagi, Sakae; Fujimaki, Satoshi; Kibayashi, Chihiro Journal of the Chemical Society, Chemical Communications, 1990 , # 20 p. 1457 - 1459 |
|
~% 
a-Homonojirimycin CAS#:119557-99-2 |
| Literature: Journal of the Chemical Society, Chemical Communications, , # 20 p. 1457 - 1459 |
|
~% 
a-Homonojirimycin CAS#:119557-99-2 |
| Literature: Journal of the Chemical Society, Chemical Communications, , # 20 p. 1457 - 1459 |
|
~% 
a-Homonojirimycin CAS#:119557-99-2 |
| Literature: Journal of the Chemical Society, Chemical Communications, , # 20 p. 1457 - 1459 |
|
~% 
a-Homonojirimycin CAS#:119557-99-2 |
| Literature: Journal of Organic Chemistry, , vol. 54, # 11 p. 2539 - 2542 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
|
In vitro inhibition of glycogen-degrading enzymes and glycosidases by six-membered sugar mimics and their evaluation in cell cultures
Bioorg. Chem. 16 , 7330-7336, (2008) An amyio-1, 6-glucosidase inhibitor enhanced inhibition of hepatic glucose production in combination with glycogen phosphorylase inhibitor. The inhibitory activity of 1-deoxynojirimycin ( 1) toward hu... |
|
|
Homonojirimycin analogues and their glucosides from Lobelia sessilifolia and Adenophora spp. (Campanulaceae).
Carbohydr. Res. 323(1-4) , 73-80, (2000) 2,6-Dideoxy-7-O-(beta-D-glucopyranosyl) 2,6-imino-D-glycero-L-gulo- heptitol (7-O-beta-D-glucopyranosyl-alpha-homonojirimycin, 1) was isolated from the 50% methanol extract of the whole plant of Lobel... |
|
|
Eight stereoisomers of homonojirimycin from D-mannose.
Org. Lett. 14(8) , 2050-3, (2012) Although there are 32 6-azidoheptitols, there are only 16 homonojirimycin (HNJ) stereoisomers. Two epimeric azidoalditols derived from d-mannose allow the synthesis in water of eight stereoisomers of ... |
| Hnj cpd |
| 3,4,5-Piperidinetriol, 2,6-bis(hydroxymethyl)-, (2α,3α,4β,5α,6β)- |
| (2r,3r,5s,6r)-2,6-bis(hydroxymethyl)piperidine-3,4,5-triol |
| (2R,3S,4R,5R,6R)-2,6-bis(hydroxymethyl)piperidine-3,4,5-triol |
| MFCD06797112 |
| Homonojirimycin |
| (2R,3R,4R,5S,6R)-2,6-Bis(hydroxymethyl)piperidine-3,4,5-triol |
| 2,6-Dideoxy-2,6-imino-D-glycero-L-gulo-heptitol |
| a-Homonojirimycin |
| α-Homonojirimycin |
| (2R,3R,4R,5S,6R)-2,6-Bis(hydroxymethyl)-3,4,5-piperidinetriol |
![(3aS,4R,6R,7R,7aS)-4-(((tert-butyldimethylsilyl)oxy)methyl)-7-(methoxymethoxy)-6-((methoxymethoxy)methyl)-2,2-dimethylhexahydro-[1,3]dioxolo[4,5-c]pyridine structure](https://www.chemsrc.com/caspic/001/132584-27-1.png)