4-(bromomethyl)phenyl acetic acid
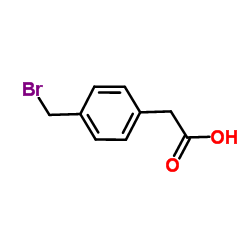
4-(bromomethyl)phenyl acetic acid structure
|
Common Name | 4-(bromomethyl)phenyl acetic acid | ||
|---|---|---|---|---|
| CAS Number | 13737-36-5 | Molecular Weight | 229.071 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 343.1±22.0 °C at 760 mmHg | |
| Molecular Formula | C9H9BrO2 | Melting Point | 179-183 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 161.3±22.3 °C | |
| Symbol |


GHS07, GHS08 |
Signal Word | Danger | |
| Name | 4-(Bromomethyl)Phenylacetic Acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 343.1±22.0 °C at 760 mmHg |
| Melting Point | 179-183 °C(lit.) |
| Molecular Formula | C9H9BrO2 |
| Molecular Weight | 229.071 |
| Flash Point | 161.3±22.3 °C |
| Exact Mass | 227.978592 |
| PSA | 37.30000 |
| LogP | 2.21 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.599 |
| Storage condition | Store at RT. |
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H315-H319-H334-H335 |
| Precautionary Statements | P261-P305 + P351 + P338-P342 + P311 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38;R42 |
| Safety Phrases | S26-S36 |
| RIDADR | UN3261 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 8 |
| HS Code | 2916399090 |
|
~72% 
4-(bromomethyl)... CAS#:13737-36-5 |
| Literature: Takahashi, Sho; Hibino, Takeshi; Sawada, Seiji Bulletin of the Chemical Society of Japan, 1988 , vol. 61, # 7 p. 2467 - 2472 |
|
~50% 
4-(bromomethyl)... CAS#:13737-36-5 |
| Literature: Bristol-Myers Company Patent: US4272437 A1, 1981 ; |
|
~% 
4-(bromomethyl)... CAS#:13737-36-5 |
| Literature: US6258954 B1, ; |
| Precursor 3 | |
|---|---|
| DownStream 10 | |
| HS Code | 2916399090 |
|---|---|
| Summary | 2916399090 other aromatic monocarboxylic acids, their anhydrides, halides, peroxides, peroxyacids and their derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:30.0% |
|
J. Chem. Soc. Chem. Commun. , 399, (1993)
|
|
|
Effect of conformational mobility and hydrogen-bonding interactions on the selectivity of some guanidinoaryl-substituted mechanism-based inhibitors of trypsin-like serine proteases.
J. Med. Chem. 35 , 4297, (1992) Previously, we have reported that some guanidino-substituted alpha- and beta-aryl enol lactones I and II behaved as selective, mechanism-based inhibitors of some trypsin-like proteases (Rai, R.; Katze... |
|
|
Synthesis and anti-inflammatory activity of the major metabolites of imrecoxib.
Bioorg. Med. Chem. Lett. 19(8) , 2270-2, (2009) We have developed a novel and moderately selective COX-2 inhibitor, imrecoxib, as a new anti-inflammatory drug. We describe herein the preparation of the major metabolites M2 and M4 of imrecoxib, as w... |
| 4-(Carboxymethyl)benzyl Bromide |
| 2-[4-(bromomethyl)phenyl]acetic acid |
| MFCD00010632 |
| 4-(BROMOMETHYL)PHENYLACETIC ACID |
| Benzeneacetic acid, 4-(bromomethyl)- |
| [4-(Bromomethyl)phenyl]acetic acid |
| 4-(bromomethyl)phenyl acetic acid |
 CAS#:176504-01-1
CAS#:176504-01-1![Diethyl 2-[4-(2,2-Dicarboethoxypropyl)phenyl]-2-methyl Malonate structure](https://www.chemsrc.com/caspic/319/189287-72-7.png) CAS#:189287-72-7
CAS#:189287-72-7![methyl 2-[4-(cyanomethyl)phenyl]acetate structure](https://www.chemsrc.com/caspic/077/193290-61-8.png) CAS#:193290-61-8
CAS#:193290-61-8 CAS#:343880-24-0
CAS#:343880-24-0 CAS#:96524-70-8
CAS#:96524-70-8 CAS#:223123-57-7
CAS#:223123-57-7 CAS#:223123-63-5
CAS#:223123-63-5 CAS#:73401-74-8
CAS#:73401-74-8![Methyl [4-(bromomethyl)phenyl]acetate structure](https://www.chemsrc.com/caspic/010/7398-42-7.png) CAS#:7398-42-7
CAS#:7398-42-7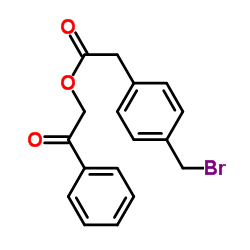 CAS#:66270-97-1
CAS#:66270-97-1
