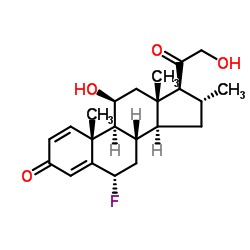
CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
TU3832500
-
CHEMICAL NAME :
-
Pregna-1,4-diene-3,20-dione, 6-alpha-fluoro-11-beta,21-dihydroxy-16-alpha-methyl-
-
CAS REGISTRY NUMBER :
-
152-97-6
-
LAST UPDATED :
-
199612
-
DATA ITEMS CITED :
-
6
-
MOLECULAR FORMULA :
-
C22-H29-F-O4
-
MOLECULAR WEIGHT :
-
376.51
-
WISWESSER LINE NOTATION :
-
L E5 B666 OV AHTTT&J A1 CQ E1 FV1Q G1 LF
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
70 mg/kg
-
SEX/DURATION :
-
female 12 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetal death
-
REFERENCE :
-
YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14, 1968-81. Suspended. Volume(issue)/page/year: 9,1623,1976
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
20 mg/kg
-
SEX/DURATION :
-
female 12 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
REFERENCE :
-
YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14, 1968-81. Suspended. Volume(issue)/page/year: 9,1623,1976
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
70 mg/kg
-
SEX/DURATION :
-
female 13 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue)
-
REFERENCE :
-
YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14, 1968-81. Suspended. Volume(issue)/page/year: 9,1623,1976
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
70 mg/kg
-
SEX/DURATION :
-
female 13 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue)
-
REFERENCE :
-
YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14, 1968-81. Suspended. Volume(issue)/page/year: 9,1623,1976
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Administration onto the skin
-
DOSE :
-
2500 ug/kg
-
SEX/DURATION :
-
female 6-15 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or resorbed implants per total number of implants)
-
REFERENCE :
-
AMBED4 AMI-Berichte. (Berlin, Fed. Rep. Ger.) No. 1-2, 1978-82. Volume(issue)/page/year: (1),127,1988
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
30 mg/kg
-
SEX/DURATION :
-
female 7-18 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus) Reproductive - Specific Developmental Abnormalities - Central Nervous System Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue)
-
REFERENCE :
-
YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14, 1968-81. Suspended. Volume(issue)/page/year: 10,151,1977
|