920022-47-5
| Name | RG7314 |
|---|---|
| Synonyms |
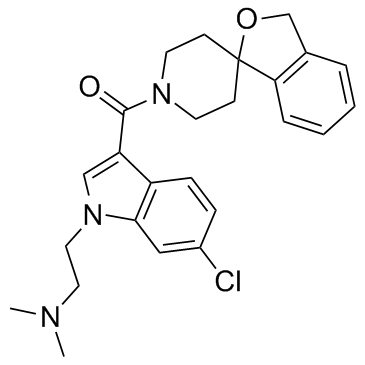
Methanone, [6-chloro-1-[2-(dimethylamino)ethyl]-1H-indol-3-yl]spiro[isobenzofuran-1(3H),4'-piperidin]-1'-yl-
RO-5028442 UNII:LZ0EU1YHCK RG7314 {6-Chloro-1-[2-(dimethylamino)ethyl]-1H-indol-3-yl}(1'H,3H-spiro[2-benzofuran-1,4'-piperidin]-1'-yl)methanone RO 5028442 RO5028442 |
| Description | RO5028442 is a highly potent and selective Brain-Penetrant Vasopressin 1a (V1a) receptor antagonist with Kis of 1 nM (hV1a) and 39 nM (mV1a). |
|---|---|
| Related Catalog | |
| Target |
Ki: 1 nM (hV1a receptor), 39 nM (mV1a receptor)[1] |
| In Vitro | RO5028442 (compound 8) has excellent binding in and functional affinity for hV1a, moderate mouse affinity, and excellent selectivity versus human V2 (hV2) and human oxytocin (hOT) receptors. RO5028442 shows high solubility. RO5028442 is found to be highly selective against a panel of 89 targets. Finally, RO5028442 is identified as a suitable compound for clinical studies[1]. |
| Cell Assay | CHO cells are stably transfected with expression plasmids encoding human V1a and grown in F-12 K, containing 10% fetal bovine serum, 1% penicillin-streptomycin, 1% glutamate, and 200 μg/mL geneticin at 37 °C in a 10% CO2 incubator at 95% humidity. Cells are plated for 24 h at 50,000 cells/well in clear bottomed 96 well plates and are dye loaded for 60 min with 2 μM Fluo-4-AM in assay buffer. After cell washing, the plate is loaded on a fluorometricimaging plate reader, compound dilution series added to the cells, and agonist activity measured[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 621.3±55.0 °C at 760 mmHg |
| Molecular Formula | C25H28ClN3O2 |
| Molecular Weight | 437.962 |
| Flash Point | 329.6±31.5 °C |
| Exact Mass | 437.187012 |
| LogP | 3.68 |
| Appearance | white solid |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.646 |
| Storage condition | -20℃ |