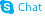189261-10-7
| Name | Natalizumab |
|---|
| Description | Natalizumab is a recombinant, humanized monoclonal antibody, binds to α4β1-integrin and blocks its interaction with vascular cell adhesion molecule-1 (VCAM-1). Natalizumab can be used for the treatment of relapsing remitting multiple sclerosis and Crohn's disease. Natalizumab is also the first targeted therapy which blocks an essential mechanism for lymphocyte entry to the CNS and thus prevents acute demyelinating relapses[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Natalizumab, a recombinant, humanized antibody, binds to α4β1-integrin and blocks its interaction with VCAM-1. As a result, leukocyte migration into brain tissue is inhibited, reducing inflammation and preventing the formation of lesions. Natalizumab may also inhibit ongoing central nervous system (CNS) inflammation, mediated by leukocytes already present in the CNS, by interrupting the interactions between α4-integrin-expressing leukocytes and extracellular matrix proteins such as fibronectin and osteopontin[1]. |
| In Vivo | Natalizumab binds rapidly and with high affinity to α4-integrin. Maximal binding (≥80% saturation), measured in vitro on isolated lymphocyte membranes, occurred 24 hours after intravenous (IV) doses of natalizumab 1 mg/kg to 6 mg/kg[1]. |
| References |
| Boiling Point | 1231.4±75.0 °C at 760 mmHg |
|---|---|
| Molecular Formula | C46H80N14O16SSe |
| Molecular Weight | 1196.237 |
| Flash Point | 698.5±37.1 °C |
| Exact Mass | 1196.476318 |
| LogP | 5.86 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |