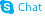851220-85-4
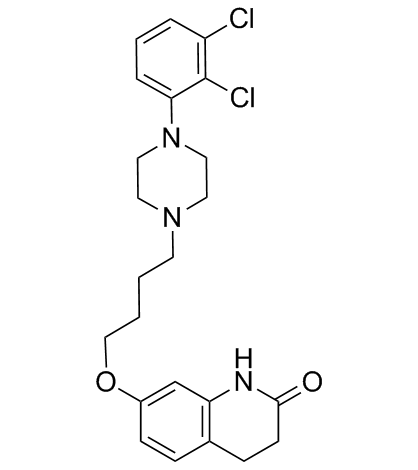
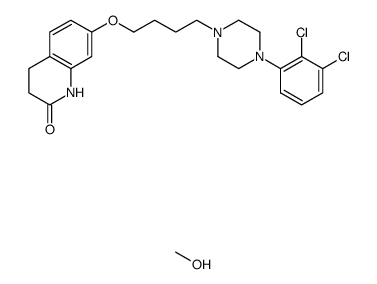
| Name | 7-[4-[4-(2,3-dichlorophenyl)piperazin-1-yl]butoxy]-3,4-dihydro-1H-quinolin-2-one,hydrate |
|---|---|
| Synonyms |
7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydro-2(1H)-quinolinone hydrate
7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydrocarbostyril monohydrate Aripiprazole monohydrate aripiprazole hydrate Aripiprazole hydrate 7-(4-[4-(2,3-dichloropheny)-1-piperazinyl]-butoxy)-3,4-dihydrocarbostyril monohydrate 7-(4-[4-(2,3-dichloropheny)-1-piperazinyl]-butoxy)-3,4-dihydro-2(1H)-quinolone monohydrate |
| Description | Aripiprazole (OPC-14597) monohydrate, an atypical antipsychotic, is a potent and high-affinity dopamine D2 receptor partial agonist. Aripiprazole monohydrate is an inverse agonist at 5-HT2B and 5-HT2A receptors and displays partial agonist actions at 5-HT1A, 5-HT2C, D3, and D4 receptors. Aripiprazole monohydrate can be used for the research of schizophrenia and COVID19[1][2][3][4]. |
|---|---|
| Related Catalog | |
| Target |
5-HT1A Receptor 5-HT2A Receptor 5-HT2B Receptor 5-HT2C Receptor D2 Receptor D3 Receptor D4 Receptor |
| In Vitro | Aripiprazole monohydrate potently activates D2 receptor-mediated inhibition of cAMP accumulation[1]. Aripiprazole monohydrate shows a greater anti-inflammatory effect on TNF-α, IL-13, IL-17α and fractalkine[3]. |
| In Vivo | Aripiprazole monohydrate (0-3 mg/kg, IP, daily) shows some anxiolytic properties[4]. Animal Model: WAG/Rij rats (N = 6 per dose, 6 months, administration of a mixture of tiletamine/zolazepam)[4] Dosage: 0, 0.3, 1, 3 mg/kg Administration: IP, 1 mL/kg, every day at 5 p.m. until the end of the experiments Result: Showed some anxiolytic properties with the 1 mg/kg dose being the most active. |
| References |
| Molecular Formula | C23H29Cl2N3O3 |
|---|---|
| Molecular Weight | 466.40100 |
| Exact Mass | 465.15900 |
| PSA | 57.53000 |
| LogP | 4.88300 |
|
~96% 
851220-85-4 |
| Literature: BRISTOL-MYERS SQUIBB COMPANY Patent: WO2005/41970 A1, 2005 ; Location in patent: Page/Page column 7; 14 ; |
|
~% 
851220-85-4 |
| Literature: SYNTHON B.V. Patent: WO2006/53780 A1, 2006 ; Location in patent: Page/Page column 23 ; |