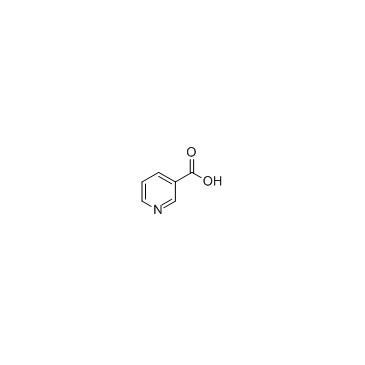
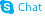29876-14-0
| Name | N-[2-(1H-indol-3-yl)ethyl]pyridine-3-carboxamide |
|---|---|
| Synonyms |
nicotredole
3-Pyridinecarboxamide, N-[2-(1H-indol-3-yl)ethyl]- Tryptamide N-Nicotinoyltryptamide N-[2-(1H-Indol-3-yl)ethyl]nicotinamide |
| Description | Nicotredole (Tryptamide) is an orally active anti-inflammatory and analgesic agent. Nicotredole exhibits evident antiinflammatory effects of potency comparable with Phenylbutazone. Nicotredole has only weak ulcerogenic activity[1][2][3]. |
|---|---|
| Related Catalog | |
| In Vitro | Nicotredole inhibits prostaglandin synthetase activity in vitro[1]. |
| In Vivo | Nicotredole produces antiinflammatory effects in carrageenin-induced rat paw oedema[1]. Nicotredole reverses pyrogen-induced hyperthermia in rats, elicits analgesic effects in rats, prolongs the time of hexobarbital sleep in rats and inhibits locomotor activity in rats and mice[1]. Nicotredole (i.p.) has LD50s of 1260 mg/kg and 1980 mg/kg for male rats and male mice, respectively[1]. Nicotredole (p.o.) has LD50s of 8.5 g/kg and 9.3 g/kg for male rats and male mice, respectively[1]. Nicotredole (25 mg/kg; p.o. or i.p.) undergoes fast absorption (t1/2=4.92-17.5 min) and elimination (t1/2=55.72-74.52 min), can reach Cmax (11-13 μg/cm3) after 30 min, and gives AUC values in the range of 21.40-27.30 (μg•h/cm3)[2]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 588.4±35.0 °C at 760 mmHg |
| Molecular Formula | C16H15N3O |
| Molecular Weight | 265.310 |
| Flash Point | 309.7±25.9 °C |
| Exact Mass | 265.121521 |
| PSA | 57.78000 |
| LogP | 1.94 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.669 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | Xi |
|---|
|
~% 
29876-14-0 |
| Literature: Journal of the American Chemical Society, , vol. 126, # 40 p. 12897 - 12906 |
|
~60% 
29876-14-0 |
| Literature: Rodriguez-Lugo, Rafael E.; Trincado, Monica; Gruetzmacher, Hansjoerg ChemCatChem, 2013 , vol. 5, # 5 p. 1079 - 1083 |
|
~81% 
29876-14-0 |
| Literature: Bodor; Farag; Polgar Journal of Pharmacy and Pharmacology, 2001 , vol. 53, # 6 p. 889 - 894 |
|
~62% 
29876-14-0 |
| Literature: Lo, Kenneth Kam-Wing; Tsang, Keith Hing-Kit; Hui, Wai-Ki; Zhu, Nianyong Chemical Communications, 2003 , # 21 p. 2704 - 2705 |
|
~% 
29876-14-0 |
| Literature: Canadian Journal of Research, Section B: Chemical Sciences, , vol. 24, p. 224,229 |
| Precursor 6 | |
|---|---|
| DownStream 0 | |