Sodium risedronate
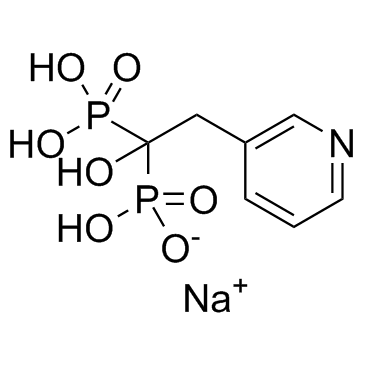
Sodium risedronate structure
|
Common Name | Sodium risedronate | ||
|---|---|---|---|---|
| CAS Number | 115436-72-1 | Molecular Weight | 305.094 | |
| Density | N/A | Boiling Point | 692.3ºC at 760 mmHg | |
| Molecular Formula | C7H10NNaO7P2 | Melting Point | 252-262°C | |
| MSDS | Chinese | Flash Point | 372.5ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Sodium risedronateRisedronate sodium is a pyridinyl biphosphonate which inhibits osteoclast-mediated bone resorption.Target: Risedronate sodium, which was promoted in Croatia a few months ago, is the latest (III) generation of bisphosphonates, the most efficient anti-resorption drugs that inhibit osteoclast-mediated bone resorption and change the bone metabolism. Risedronate sodium is hence the first line of bisphosphonates for the reduction of vertebral and non-vertebral fracture risks in postmenopausal women with osteoporosis or those with a high risk of osteoporosis. It also efficiently prevents bone loss or improves bone density in men and women on a long-term corticosteroid therapy [1].The administration of 20 and 25 mg/kg risedronate sodium for 4 days led to decreases of parasitemia of 68.9% and 83.6%, respectively. On the seventh day of treatment the inhibitions were 63% and 88.9% with 20 and 25 mg/kg, respectively. After recovering the parasitemia, a dose-response curve was obtained for estimating the ID50 (dose causing 50% inhibition), equivalent to 17 ± 1.8 mg/kg after 7 days of treatment. Four days after the interruption of treatment (11 days postinfection), the parasitemias of the groups treated with 10, 15, 20, and 25 mg/kg/day were 15.3%, 15.9%, 15.2%, and 5.7%, respectively. Conversely, the group that received PBS presented parasitemia of 25.6%. Among the groups treated with risedronate sodium, only the animals that received 25 mg/kg had a significant inhibition of 77.8% (see Table S1 in the supplemental material), demonstrating that even after treatment discontinuation, the parasitemia of the animals remained low in relation to that of the controls [2].Clinical indications: Bone resorption; Male osteoporosis; Osteogenesis imperfecta; Osteoporosis; Pagets bone disease Toxicity: abdominal pain; anxiety, back pain; belching, bladder irritation; bone disorders and pain; bronchitis; bursitis; cataracts; chest pain; colitis; constipation; depression; diarrhea; difficulty breathing |
| Name | Sodium risedronate |
|---|---|
| Synonym | More Synonyms |
| Description | Risedronate sodium is a pyridinyl biphosphonate which inhibits osteoclast-mediated bone resorption.Target: Risedronate sodium, which was promoted in Croatia a few months ago, is the latest (III) generation of bisphosphonates, the most efficient anti-resorption drugs that inhibit osteoclast-mediated bone resorption and change the bone metabolism. Risedronate sodium is hence the first line of bisphosphonates for the reduction of vertebral and non-vertebral fracture risks in postmenopausal women with osteoporosis or those with a high risk of osteoporosis. It also efficiently prevents bone loss or improves bone density in men and women on a long-term corticosteroid therapy [1].The administration of 20 and 25 mg/kg risedronate sodium for 4 days led to decreases of parasitemia of 68.9% and 83.6%, respectively. On the seventh day of treatment the inhibitions were 63% and 88.9% with 20 and 25 mg/kg, respectively. After recovering the parasitemia, a dose-response curve was obtained for estimating the ID50 (dose causing 50% inhibition), equivalent to 17 ± 1.8 mg/kg after 7 days of treatment. Four days after the interruption of treatment (11 days postinfection), the parasitemias of the groups treated with 10, 15, 20, and 25 mg/kg/day were 15.3%, 15.9%, 15.2%, and 5.7%, respectively. Conversely, the group that received PBS presented parasitemia of 25.6%. Among the groups treated with risedronate sodium, only the animals that received 25 mg/kg had a significant inhibition of 77.8% (see Table S1 in the supplemental material), demonstrating that even after treatment discontinuation, the parasitemia of the animals remained low in relation to that of the controls [2].Clinical indications: Bone resorption; Male osteoporosis; Osteogenesis imperfecta; Osteoporosis; Pagets bone disease Toxicity: abdominal pain; anxiety, back pain; belching, bladder irritation; bone disorders and pain; bronchitis; bursitis; cataracts; chest pain; colitis; constipation; depression; diarrhea; difficulty breathing |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 692.3ºC at 760 mmHg |
|---|---|
| Melting Point | 252-262°C |
| Molecular Formula | C7H10NNaO7P2 |
| Molecular Weight | 305.094 |
| Flash Point | 372.5ºC |
| Exact Mass | 304.983032 |
| PSA | 170.63000 |
| LogP | 0.06380 |
| Vapour Pressure | 4.03E-20mmHg at 25°C |
| Storage condition | Store at RT |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 + H312 + H332 |
| Precautionary Statements | P280 |
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| RTECS | SZ8567500 |
|
Ionic complex of risedronate with positively charged deoxycholic acid derivative: evaluation of physicochemical properties and enhancement of intestinal absorption in rats.
Arch. Pharm. Res. 37(12) , 1560-9, (2014) Risedronate is widely used clinically to treat osteoporosis, Paget's disease, hypercalcemia, bone metastasis, and multiple myeloma. However, its oral efficacy is restricted due to its low bioavailabil... |
|
|
Effects of various squalene epoxides on coenzyme Q and cholesterol synthesis.
Biochim. Biophys. Acta 1841(7) , 977-86, (2014) 2,3-Oxidosqualene is an intermediate in cholesterol biosynthesis and 2,3:22,23-dioxidosqualene act as the substrate for an alternative pathway that produces 24(S),25-epoxycholesterol which effects cho... |
|
|
Risedronate increases osteoblastic differentiation and function through connexin43.
Biochem. Biophys. Res. Commun. 432(1) , 152-6, (2013) Bisphosphonates are potent antiresorptive drugs which have antifracture efficacy by reducing bone turnover rate and increasing bone mineral density. In addition to inhibiting osteoclast function, bisp... |
| T6NJ C1XQPQQO&PQQO &&Mono Na salt |
| Actonel |
| Natriumhydrogen-(1-hydroxy-1-phosphono-2-pyridin-3-ylethyl)phosphonat |
| Risedronic acid monosodium salt |
| sodium hydrogen (1-hydroxy-1-phosphono-2-pyridin-3-ylethyl)phosphonate |
| Benet |
| hydrogène (1-hydroxy-1-phosphono-2-pyridin-3-yléthyl)phosphonate de sodium |
| Phosphonic acid, [1-hydroxy-2-(3-pyridinyl)ethylidene]bis-, sodium salt (1:1) |
| MFCD01706268 |
| phosphonic acid, [1-hydroxy-2-(3-pyridinyl)ethylidene]bis-, monosodium salt |
| Sodium risedronate |
| sodium hydrogen [1-hydroxy-1-phosphono-2-(pyridin-3-yl)ethyl]phosphonate |
| P,P'-[1-Hydroxy-2-(3-pyridonyl)ethylidene]bis-phosphonic acid sodium salt |
| Sodium hydrogen [1-hydroxy-1-phosphono-2-(3-pyridinyl)ethyl]phosphonate |
| Risedronic acid sodium salt |
| sodium hydrogen-1-hydroxy-1-phosphono-2-(pyridin-3-yl)ethylphosphonate |
| [1-Hydroxy-2-(3-pyridinyl)ethylidene]bis(phosphonic acid), sodium salt (1:1) |
| Optinate |
| Risedronate sodium |