Azithromycin Dihydrate
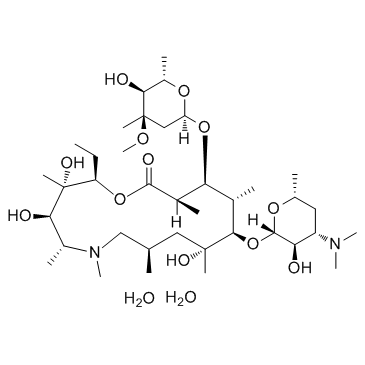
Azithromycin Dihydrate structure
|
Common Name | Azithromycin Dihydrate | ||
|---|---|---|---|---|
| CAS Number | 117772-70-0 | Molecular Weight | 785.015 | |
| Density | 1.18g/cm3 | Boiling Point | 822.1ºC at 760mmHg | |
| Molecular Formula | C38H76N2O14 | Melting Point | 113-115ºC | |
| MSDS | Chinese USA | Flash Point | 451ºC | |
Use of Azithromycin DihydrateAzithromycin hydrate is a macrolide antibiotic useful for the treatment of a number of bacterial infections. |
| Name | azithromycin dihydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Azithromycin hydrate is a macrolide antibiotic useful for the treatment of a number of bacterial infections. |
|---|---|
| Related Catalog | |
| In Vitro | Azithromycin (2 μM) augments rhinovirus-induced IFNβ expression in primary bronchial epithelial cells from asthmatics, which is associated with over-expression of RIG-I like receptors and repression of viral replication. Knockdown of MDA5, but not knockdown of RIG-I, diminishes azithromycin (2 μM)-enhanced viral-induced IFNβ expression in asthmatic primary bronchial epithelial cells[1]. Azithromycin specifically reduces MMP-9 mRNA and protein levels without affecting NF-κB in endotoxin-challenged monocytic THP-1 cells[2]. |
| In Vivo | Azithromycin (50 mg/kg) has no effect on bronchoalveolar lavage inflammatory parameters and LDH levels in a mouse model of asthma exacerbation. Azithromycin induces neither general inflammatory parameters nor LDH release in a mouse model of asthma exacerbation, and augments expression of interferon-stimulated genes and the pattern recognition receptor MDA5 but not RIG-I in exacerbating mice[1]. |
| Cell Assay | THP-1 cells (106 cells in 1 mL RPMI medium, without antibiotics, growth factors or serum) are seeded in each well of 24-well plates and allowed to settle for 1 hour. Next, 50 μL of the test compound is added followed by 50 μL of LPS (final concentration of 10 μg/mL). After 24h (37°C and 5% CO2) the supernatants and cell pellets are collected (1200 rpm, 5 min). THP-1 cell viability is tested using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT). MTT is dissolved at 2 mg/mL in PBS and aliquots are stored at -20°C. The MTT assay is performed according to the suppliers instructions. Absorbance of MTT converted into formazan is measured at a wavelength of 570 nm with background subtraction at 630 nm. |
| References |
| Density | 1.18g/cm3 |
|---|---|
| Boiling Point | 822.1ºC at 760mmHg |
| Melting Point | 113-115ºC |
| Molecular Formula | C38H76N2O14 |
| Molecular Weight | 785.015 |
| Flash Point | 451ºC |
| Exact Mass | 784.529663 |
| PSA | 198.54000 |
| LogP | 1.71000 |
| Vapour Pressure | 2.51E-31mmHg at 25°C |
| Hazard Codes | Xi |
|---|---|
| Risk Phrases | R36/37/38 |
| Safety Phrases | 24/25-36/37/39 |
| RIDADR | NONH for all modes of transport |
| RTECS | RN6960000 |
| HS Code | 29419000 |
|
Environmental friendly method for urban wastewater monitoring of micropollutants defined in the Directive 2013/39/EU and Decision 2015/495/EU.
J. Chromatogr. A. 1418 , 140-9, (2015) The fate and removal of organic micropollutants in the environment is a demanding issue evidenced by the recent European policy. This work presents an analytical method for the trace quantification of... |
|
|
Purification and characterization of the Staphylococcus aureus bacillithiol transferase BstA.
Biochim. Biophys. Acta 1840(9) , 2851-61, (2014) Gram-positive bacteria in the phylum Firmicutes synthesize the low molecular weight thiol bacillithiol rather than glutathione or mycothiol. The bacillithiol transferase YfiT from Bacillus subtilis wa... |
|
|
A comparison of in vitro ADME properties and pharmacokinetics of azithromycin and selected 15-membered ring macrolides in rodents.
Eur. J. Drug Metab. Pharmacokinet. 39(4) , 263-76, (2014) The purpose of this study was to evaluate the impact of structural modifications on the 15-membered macrolactone ring and/or substituents on the in vitro ADME properties and in vivo pharmacokinetic (P... |
| (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-2-Ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-15-oxo-11-{[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy}-1-oxa-6-azacyclopentadecan-13-yl 2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranoside dihydrate |
| 1-Oxa-6-azacyclopentadecan-15-one |
| 1-Oxa-6-azacyclopentadecan-15-one, 13-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-11-[[3,4,6-trideoxy-3-(dimethylamino)-b η-D-xylo-hexopyranosyl]oxy]-, (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-, hydrate (1:2) |
| N-Methyl-11-aza-10-deoxo-10-dihydroerythromycin A CP-62993 |
| Azithromycindihydrate |
| 1-oxa-6-azacyclopentadecan-15-one, 13-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-11-[[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy]-, (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-, hydrate (1:2) |
| Azithromycin Dihydrate |
| Azithromycin (hydrate) |

