Naproxen
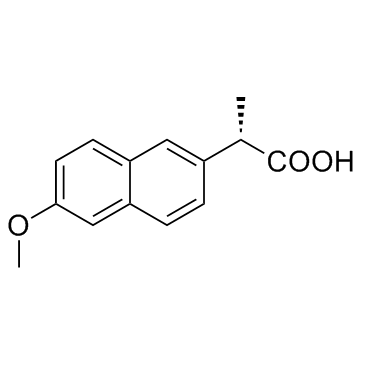
Naproxen structure
|
Common Name | Naproxen | ||
|---|---|---|---|---|
| CAS Number | 22204-53-1 | Molecular Weight | 230.259 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 403.9±20.0 °C at 760 mmHg | |
| Molecular Formula | C14H14O3 | Melting Point | 152-154 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 154.5±15.3 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of NaproxenNaproxen is a COX-1 and COX-2 inhibitor with IC50s of 8.72 and 5.15 μM, respectively in cell assay. |
| Name | naproxen |
|---|---|
| Synonym | More Synonyms |
| Description | Naproxen is a COX-1 and COX-2 inhibitor with IC50s of 8.72 and 5.15 μM, respectively in cell assay. |
|---|---|
| Related Catalog | |
| Target |
COX-2:5.65 μM (IC50) COX-1:9.55 μM (IC50) |
| In Vitro | Naproxen etemesil is a lipophilic, non-acidic, inactive prodrug of naproxen that is hydrolysed to pharmacologically active Naproxen once absorbed. Naproxen is a well known nonsteroidal anti-inflammatory drug. Naproxen is approximately equipotent inhibitor of COX-1 and COX-2 in intact cells with IC50s of 2.2 μg/mL and 1.3 μg/mL, respectively[1]. |
| In Vivo | Naproxen exerts an anti-inflammatory and antifibrotic effect in mouse model of bleomycin-induced lung fibrosis. Naproxen also downregulates TGF-β levels and Smad3/4 complex formation[2]. Naproxen is shown to inhibit the time-courses of pain, fever and PGE2 with similar potencies (IC50=27, 40, 13 μM)[3]. |
| Cell Assay | BAEC are incubated for 30 min with Naproxen (0.1 ng/mL to 1 mg/mL). Arachidonic acid (30 μM) is then added, and the cells are incubated for a further 15 min at 37°C. The medium is then removed, and radioimmunoassay is used to measure the formation of 6-keto-PGF,a, PGE2, thromboxane B2, or PGF2a[1]. |
| Animal Admin | Rats[3] To measure the analgesic effects of naproxen in a carrageenaninduced model of monoarthritis, Male Sprague–Dawley rats (n=48, 217±28 g) are randomly divided into four groups of 12 by an internally developed computer program, allowing the blind performance of the behavioral experiment. To induce hyperalgesia by inflammation, animals in groups 1B, 1C, and 1D receive a 40-μL intra-articular injection of a saline solution containing 7.5 mg/mL carrageenan in the left hind limb under isoflurane anesthesia (time=−1 h). Animals in group 1A receive no injection. After 1 h (time=0) the animals in groups 1A, 1B, 1C, and 1D receive oral doses of naproxen in saline of 0, 0, 7.5 and 30 μmol/kg, respectively. The doses and time points of measurements are selected on the basis of simulations predicting measuring a full concentration-effect relationship within the time-span of the experiment[3]. Mice[2] Bleomycin (0.05 IU) is instilled intratracheally to C57BL/6 mice, which are then treated by micro-osmotic pump with vehicle, JNJ7777120 (40 mg/kg b.wt.), naproxen (21 mg/kg b.wt.), or a combination of both. Airway resistance to inflation, an index of lung stiffness, is assessed, and lung specimens are processed for inflammation, oxidative stress, and fibrosis markers[2]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 403.9±20.0 °C at 760 mmHg |
| Melting Point | 152-154 °C(lit.) |
| Molecular Formula | C14H14O3 |
| Molecular Weight | 230.259 |
| Flash Point | 154.5±15.3 °C |
| Exact Mass | 230.094299 |
| PSA | 46.53000 |
| LogP | 3.00 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.609 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R22 |
| Safety Phrases | S36/37 |
| RIDADR | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | UF5275000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2916399090 |
|
~81% 
Naproxen CAS#:22204-53-1 |
| Literature: Nagasawa, Kazuo; Yoshitake, Shinji; Amiya, Takao; Ito, Keiichi Synthetic Communications, 1990 , vol. 20, # 13 p. 2033 - 2040 |
|
~% 
Naproxen CAS#:22204-53-1 |
| Literature: Harrison; Lewis; Nelson; Rooks; Roszkowski; Tomolonis; Fried Journal of medicinal chemistry, 1970 , vol. 13, # 2 p. 203 - 205 |
|
~% 
Naproxen CAS#:22204-53-1 |
| Literature: Harrison; Lewis; Nelson; Rooks; Roszkowski; Tomolonis; Fried Journal of medicinal chemistry, 1970 , vol. 13, # 2 p. 203 - 205 |
| Precursor 2 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918990090 |
|---|---|
| Summary | 2918990090. other carboxylic acids with additional oxygen function and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Environmental friendly method for urban wastewater monitoring of micropollutants defined in the Directive 2013/39/EU and Decision 2015/495/EU.
J. Chromatogr. A. 1418 , 140-9, (2015) The fate and removal of organic micropollutants in the environment is a demanding issue evidenced by the recent European policy. This work presents an analytical method for the trace quantification of... |
|
|
Validation of cyclooxygenase-2 as a direct anti-inflammatory target of 4-O-methylhonokiol in zymosan-induced animal models.
Arch. Pharm. Res. 38 , 813-25, (2015) 4-O-methylhonokiol (MH) is known to inhibit inflammation by partially understood mechanisms. Here, the anti-inflammatory mechanisms of MH were examined using enzymatic, cellular, and animal assays. In... |
|
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
| Equiproxen |
| piproxen |
| (+)-(S)-Naproxen |
| (2S)-2-(6-methoxynaphthalen-2-yl)propanoic acid |
| (2S)-2-(6-Methoxy-2-naphthyl)propanoic acid |
| 2-Naphthaleneacetic acid, 6-methoxy-α-methyl-, (S)- |
| 2-naphthaleneacetic acid, 6-methoxy-a-methyl-, (aS)- |
| Naproxen |
| Apronax |
| Naprosyne |
| Naproxene |
| (+)-NAPROXEN |
| (S)-(+)-6-methoxy-α-methyl-2-naphthaleneacetic acid |
| ALEVE |
| (S)-naproxen |
| Anaprox |
| (S)-(+)-2-(6-Methoxy-2-naphthyl)propionic Acid |
| Naproxeno |
| 2-Naphthaleneacetic acid, 6-methoxy-α-methyl-, (αS)- |
| NAPRELAN |
| Naproxenum |
| MFCD00010500 |
| (2S)-2-(6-Methoxynaphth-2-yl)propanoic acid |
| EINECS 244-838-7 |
| (S)-(+)-Naproxen |
| Naprosyn |

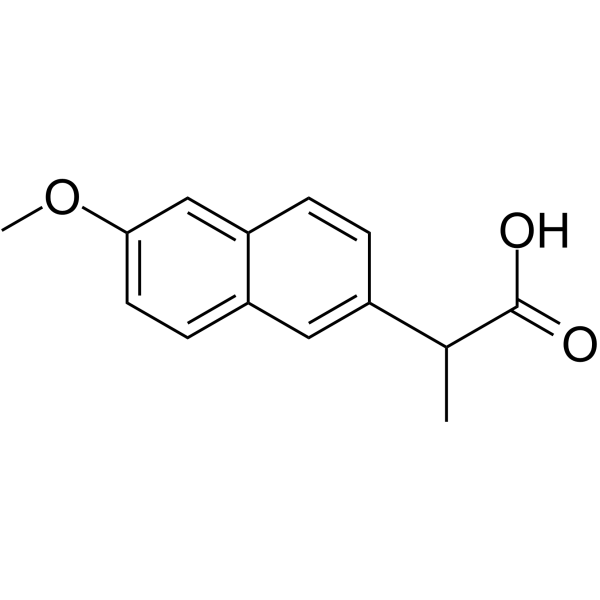
 CAS#:51091-84-0
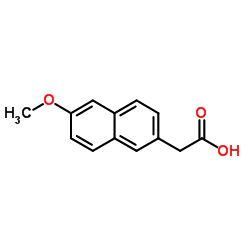
CAS#:51091-84-0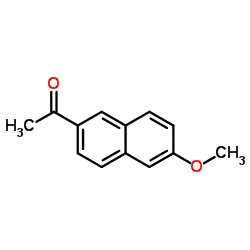 CAS#:3900-45-6
CAS#:3900-45-6 CAS#:52079-10-4
CAS#:52079-10-4 CAS#:7440-44-0
CAS#:7440-44-0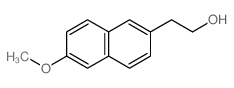 CAS#:32725-05-6
CAS#:32725-05-6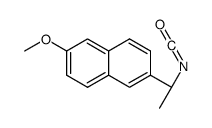 CAS#:125836-70-6
CAS#:125836-70-6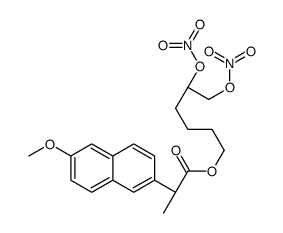 CAS#:1262956-64-8
CAS#:1262956-64-8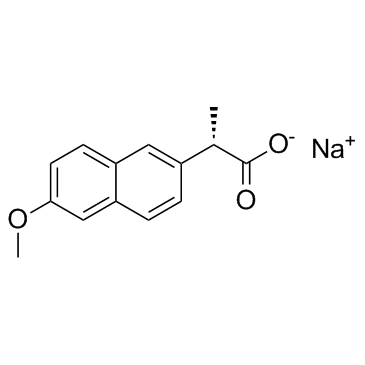 CAS#:26159-34-2
CAS#:26159-34-2 CAS#:163133-43-5
CAS#:163133-43-5
