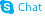1169755-45-6
| Name | (Z)-1-(3-ethyl-5-hydroxybenzo[d]thiazol-2(3H)-ylidene)propan-2-one |
|---|---|
| Synonyms | Phencyclidine hydrochloride |
| Description | INDY is a potent and ATP-competitive Dyrk1A and Dyrk1B inhibitor with IC50s of 0.24 μM and 0.23 μM, respectively. INDY binds in the ATP pocket of the enzyme and has a Ki value of 0.18 μM for Dyrk1A. INDY sharply reduces the self-renewal capacity of normal and tumorigenic cells in primary Glioblastoma (GBM) cell lines and neural progenitor cells[1][2]. |
|---|---|
| Related Catalog | |
| Target |
IC50: 0.24 μM (Dyrk1A) and 0.23 μM (Dyrk1B)[1] Ki: 0.18 μM (Dyrk1A)[1] |
| In Vitro | INDY (0.3-30 μM; 20 hours) mildly inhibits tau-phosphorylation at 3 μM, and nearly completely inhibits at 30 μM[1]. INDY effectively reverses the aberrant tau-phosphorylation and rescues the repressed NFAT (nuclear factor of activated T cell) signalling induced by Dyrk1A (dual-specificity tyrosine-(Y)-phosphorylation-regulated kinase 1A) overexpression[1]. Western Blot Analysis[1] Cell Line: COS7 cells transfected with either EGFP-Dyrk1A and EGFP-tau Concentration: 0.3, 1, 3, 10, 30 μM Incubation Time: 20 hours Result: Mildly inhibited tau-phosphorylation at 3 μM, and nearly completely inhibited at 30 μM. |
| In Vivo | ProINDY (2.5 μM) recoveres apparently normal development of the Xenopus embryo[1]. |
| References |
| Molecular Formula | C12H13NO2S |
|---|---|
| Molecular Weight | 235.30200 |
| Exact Mass | 235.06700 |
| PSA | 70.47000 |
| LogP | 2.07890 |
| Storage condition | -20°C |
| RIDADR | NONH for all modes of transport |
|---|