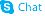155569-91-8
| Name | Emamectin benzoate |
|---|---|
| Synonyms |
(2aE,4E,5'S,6S,6'R,7S,8E,11R,13S,15S,17aR,20R,20aR,20bS)-20,20b-dihydroxy-5',6,8,19-tetramethyl-6'-[(1S)-1-methylpropyl]-17-oxo-5',6,6',10,11,14,15,17,17a,20,20a,20b-dodecahydro-2H,7H-spiro[11,15-methanofuro[4,3,2-pq][2,6]benzodioxacyclooctadecine-13,2'-pyran]-7-yl 2,6-dideoxy-3-O-methyl-4-O-[2,4,6-trideoxy-3-O-methyl-4-(methylammonio)-a-L-arabino-hexopyranosyl]-a-L-arabino-hexopyranoside benzoate
Emamectin Benzoate (1'R,2S,4'S,5S,6R,8'R,10'E,12'S,13'S,14'E,16'E,20'R,21'R,24'S)-6-[(2S)-2-Butanyl]-21',24'-dihydroxy-5,11',13',22'-tetramethyl-2'-oxo-5,6-dihydrospiro[pyran-2,6'-[3,7,19]trioxatetracyclo[15.6.1.1. 0]pentacosa[10,14,16,22]tetraen]-12'-yl 2,6-dideoxy-3-O-methyl-4-O-[2,4,6-trideoxy-3-O-methyl-4-(methylammonio)-α-L-arabino-hexopyranosyl]-α-L-arabino-hexopyranoside benzoate (2aE,4E,5'S,6S,6'R,7S,8E,11R,13S,15S,17aR,20R,20aR,20bS)-20,20b-dihydroxy-5',6,8,19-tetramethyl-6'-[(1S)-1-methylpropyl]-17-oxo-5',6,6',10,11,14,15,17,17a,20,20a,20b-dodecahydro-2H,7H-spiro[11,15-meth Emamectin (Benzoate) (4''R)-4''-Deoxy-4''-(methylamino)-avermectin B1 benzoate |
| Description | Emamectin Benzoate (MK-244) works as a chloride channel activator by binding gamma aminobutyric acid (GABA) receptor and glutamate-gated chloride channels disrupting nerve signals within arthropods. |
|---|---|
| Related Catalog | |
| Target |
GABA Receptor |
| In Vitro | Emamectin Benzoate stimulates the release of GABA from the synapses between nerve cells and while additionally increasing GABA's affinity for its receptor on the post-junction membrane of muscle cells in insects and arthropods. |
| References |
| Melting Point | 141-146ºC |
|---|---|
| Molecular Formula | C104H154N2O28 |
| Molecular Weight | 1880.33 |
| PSA | 206.57000 |
| LogP | 4.62840 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | T,N,Xi |
|---|---|
| Risk Phrases | 25-36-50-57-58-36/37/38-50/53-23-21 |
| Safety Phrases | 26-36-45-60-61-36/37 |
| RIDADR | UN 2811 6.1/PG 3 |
| RTECS | CL1203005 |