16469-85-5
| Name | 1,3-dimethoxy-d6-benzene |
|---|---|
| Synonyms |
1,3-bis-tert-butylsulfanyl-propane
1,3-Bis-tert-butylmercapto-propan 2,2,8,8-Tetramethyl-3,7-dithianonan Di-tert.-butylthio-1.3-propan 1.3-Bis-<tert-butyl-thio>-propan 1,3-bis-trideuteriomethoxybenzene 1,3-Bis[(H)methyloxy]benzene Benzene, 1,3-bis(methyl-d-oxy)- |
| Description | 1,3-Dimethoxybenzene-d6 is the deuterium labeled 1,3-Dimethoxybenzene[1]. 1,3-Dimethoxybenzene belongs to the class of organic compounds known as dimethoxybenzenes. 1,3-Dimethoxybenzene is an intermediate in synthesis of organic compounds[2]. |
|---|---|
| Related Catalog | |
| In Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs[1]. |
| References |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 217.5±0.0 °C at 760 mmHg |
| Molecular Formula | C8H4D6O2 |
| Molecular Weight | 144.201 |
| Flash Point | 87.8±0.0 °C |
| Exact Mass | 144.105743 |
| PSA | 18.46000 |
| LogP | 1.93 |
| Vapour Pressure | 0.2±0.4 mmHg at 25°C |
| Index of Refraction | 1.488 |
| Storage condition | 2-8°C |
|
~6% 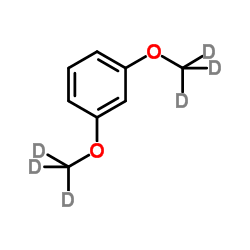
16469-85-5 |
| Literature: Fitjer, Lutz; Steeneck, Christoph; Gaini-Rahimi, Said; Schroeder, Ulrike; Justus, Karl; Puder, Peter; Dittmer, Martin; Hassler, Carla; Weiser, Juerg; Noltemeyer, Mathias; Teichert, Markus Journal of the American Chemical Society, 1998 , vol. 120, # 2 p. 317 - 328 |