Urocanic acid
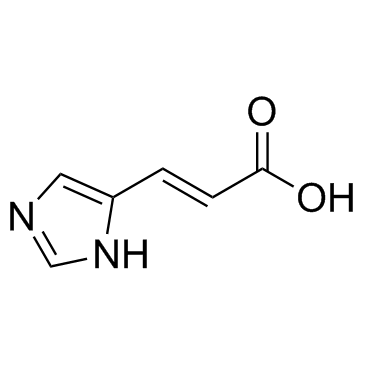
Urocanic acid structure
|
Common Name | Urocanic acid | ||
|---|---|---|---|---|
| CAS Number | 104-98-3 | Molecular Weight | 138.124 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 456.9±20.0 °C at 760 mmHg | |
| Molecular Formula | C6H6N2O2 | Melting Point | 226-228 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 230.1±21.8 °C | |
Use of Urocanic acidUrocanic acid, produced in the upper layers of mammalian skin, is a major absorber of ultraviolet radiation (UVR). |
| Name | urocanic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Urocanic acid, produced in the upper layers of mammalian skin, is a major absorber of ultraviolet radiation (UVR). |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | Urocanic acid (UCA) is formed in the upper layers of the epidermis where filaggrin, a histidine-rich filamentous protein produced after caspase-14 cleavage of profilaggrin, is broken down by proteinases into component amino acids[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 456.9±20.0 °C at 760 mmHg |
| Melting Point | 226-228 °C(lit.) |
| Molecular Formula | C6H6N2O2 |
| Molecular Weight | 138.124 |
| Flash Point | 230.1±21.8 °C |
| Exact Mass | 138.042923 |
| PSA | 65.98000 |
| LogP | 0.01 |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.674 |
| Storage condition | 2-8°C |
| Water Solubility | SLIGHTLY SOLUBLE |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NI3425200 |
| HS Code | 29332990 |
|
~% 
Urocanic acid CAS#:104-98-3 |
| Literature: Analytical Chemistry, , vol. 77, # 4 p. 1013 - 1019 |
| HS Code | 29332990 |
|---|
|
Investigation of a Degradant in a Biologics Formulation Buffer Containing L-Histidine.
Pharm. Res. 32 , 2625-35, (2015) An unknown UV 280 nm absorbing peak was observed by SEC for protein stability samples formulated in L-histidine during a stress stability study. Understanding the source would enhance the confidence i... |
|
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain canc... |
|
|
Development of imidazole alkanoic acids as mGAT3 selective GABA uptake inhibitors.
Eur. J. Med. Chem. 46 , 1483-98, (2011) A new series of potential GABA uptake inhibitors starting from of 1H-imidazol-4-ylacetic acid with the carboxylic acid side chain originating from different positions and varying in length have been s... |
| (2E)-3-(1H-Imidazol-4-yl)acrylic acid |
| urocanoic acid |
| (2E)-3-(1H-imidazol-4-yl)prop-2-enoic acid |
| (E)-3-(1H-imidazol-4-yl)acrylic acid |
| RARECHEM AL BK 0762 |
| (Z/E)-3-(1H-imidazol-4-yl)-2-propenoic acid |
| 3-(1H-imidazol-4-yl)prop-2-enoic acid |
| imidazoleacrylicacid |
| (E)-3-(1H-imidazol-4-yl)-2-propenoic acid |
| 3-imidazol-4-ylacrylic |
| Urocanate |
| 4-Imidazoleacrylic Acid |
| 2-Propenoic acid, 3-(1H-imidazol-4-yl)-, (2E)- |
| UROCANINIC ACID |
| Trans-Urocanic acid |
| (E)-3-(1H-Imidazol-5-yl)-2-propenoic acid |
| 3-(1H-Imidazol-4-yl)acrylic acid |
| Urocanic caid |
| Urocanic acid |
| 3-imidazol-4-yl-acrylic acid |
| 2-propenoic acid, 3-(1H-imidazol-5-yl)-, (2E)- |
| urosanic acid |
| 4-imidazoleacrylate |
| MFCD00005203 |
| (2E)-3-(1H-Imidazol-5-yl)acrylic acid |
| EINECS 203-258-4 |
 CAS#:31434-93-2
CAS#:31434-93-2 CAS#:102676-60-8
CAS#:102676-60-8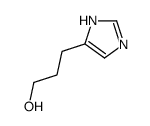 CAS#:49549-75-9
CAS#:49549-75-9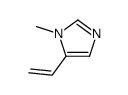 CAS#:56662-92-1
CAS#:56662-92-1
