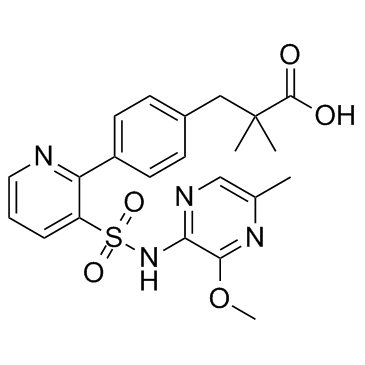
| Description |
ZD-1611 is a potent, orally active, selective ETA receptor antagonist, used for the research of ischemic stroke.
|
| Related Catalog |
|
| In Vitro |
ZD1611 competitively inhibits 125I-labeled ET-1 binding at human cloned ETA and ETB receptors with pIC50 values of 8.6 and 5.6, respectively, showing 1000-fold selectivity for the ETA receptor[1].
|
| In Vivo |
ZD1611 (0.3 mg/kg, p.o.) has a duration of action of more than 7 h in rats. In the dog, ZD1611 is active for at least 6 h at dose of 0.6 mg/kg p.o[1]. ZD1611 (0.15 mg/kg/day) in combination with candesartan decreases the brain damage and improves the neurological scores in rats. However, ZD1611 or candesartan alone does not significantly decrease the brain damage or improve neurological scores[2].
|
| Animal Admin |
The precursor of ET-1, big ET-1, is used for in vivo analysis of the effects of ZD1611. Exogenously administered big ET-1 is converted to the biologically active peptide ET-1 in vivo via a phosphoramidon-sensitive ET-converting enzyme. In the present study, the use of big ET-1 in vivo is preferred because this compound fails to elicit the initial depressor response associated with i.v. administered ET-1 and yields a greater maximum response than that to ET-1 itself. A partial cumulative dose-response curve to i.v. big ET-1 starting at 0.3 nmol/kg) is constructed until pressor responses >30 mm Hg are achieved. After a 55-min recovery period, ZD1611 (0.03-0.3 mg/kg) or vehicle is administered, and the big ET-1-response curve is repeated 5 min later. The activity of ZD1611 is calculated as a ratio of the dose of big ET-1 required to give a 30-mm Hg rise in MAP in the absence and then the presence of the compound.
|
| References |
[1]. Wilson C, et al. Pharmacological profile of ZD1611, a novel, orally active endothelin ETA receptor antagonist. J Pharmacol Exp Ther. 1999 Sep;290(3):1085-91. [2]. Stenman E, et al. Cooperative effect of angiotensin AT(1) and endothelin ET(A) receptor antagonism limits the brain damage after ischemic stroke in rat. Eur J Pharmacol. 2007 Sep 10;570(1-3):142-8. Epub 2007 Jun 9.
|