Gambogic Acid
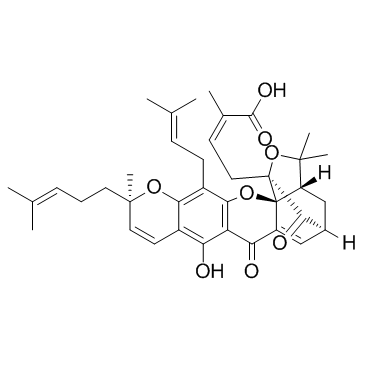
Gambogic Acid structure
|
Common Name | Gambogic Acid | ||
|---|---|---|---|---|
| CAS Number | 2752-65-0 | Molecular Weight | 628.751 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 808.9±65.0 °C at 760 mmHg | |
| Molecular Formula | C38H44O8 | Melting Point | 88.5°C | |
| MSDS | Chinese USA | Flash Point | 251.4±27.8 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of Gambogic AcidGambogic acid is derived from the gamboges resin of the tree Garcinia hanburyi. Gambogic acid inhibits Bcl-XL, Bcl-2, Bcl-W, Bcl-B, Bfl-1 and Mcl-1 with IC50s of 1.47 μM, 1.21 μM, 2.02 μM, 0.66 μM, 1.06 μM and 0.79 μM. |
| Name | gambogic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Gambogic acid is derived from the gamboges resin of the tree Garcinia hanburyi. Gambogic acid inhibits Bcl-XL, Bcl-2, Bcl-W, Bcl-B, Bfl-1 and Mcl-1 with IC50s of 1.47 μM, 1.21 μM, 2.02 μM, 0.66 μM, 1.06 μM and 0.79 μM. |
|---|---|
| Related Catalog | |
| Target |
Bcl-B:0.66 μM (IC50) Mcl-1:0.79 μM (IC50) Bfl-1:1.06 μM (IC50) Bcl-2:1.21 μM (IC50) Bcl-xL:1.47 μM (IC50) Bcl-W:2.02 μM (IC50) Autophagy |
| In Vitro | Gambogic Acid is a medicinal compound derived from the gamboges resin of the tree, Garcinia hanburyi. Gambogic Acid has documented cytotoxic activity against tumor cell lines in culture, with concentrations required for killing 50% of cells (LD50 of ~1 μM). The activity of Gambogic Acid against the 6 human anti-apoptotic Bcl-2-family proteins is contrasted, using FPAs. Gambogic Acid displaces to various extents FITC-BH3 peptide binding to all 6 proteins, with apparent IC50 1.47 μM for Bcl-XL, 1.21 μM for Bcl-2, 2.02 μM for Bcl-W, 0.66 μM for Bcl-B, 1.06 μM for Bfl-1, and 0.79 μM for Mcl-1[1]. The growth inhibitory effects of Gambogic Acid (GA) or Cisplatin (CDDP) on A549, NCI-H460, and NCI-H1299 cells are assessed by the MTT assay after 48 h exposure. A concentration-dependent inhibition of cell growth is observed with Gambogic Acid and CDDP, with IC50s of 3.56±0.36 and 21.88±3.21 μM for A549 cells, 4.05±0.51 and 25.76±4.03 μM for NCI-H460 cells, and 1.12±0.31 μM and 25.21±4.38 μM for NCI-H1299 cells[2]. |
| In Vivo | To further investigate whether Gambogic Acid synergises CDDP against tumour growth in vivo, A549 tumors are implanted in SCID mice. When mice are treated with CDDP combined with Gambogic Acid, the tumor inhibition rate is 69.3%, whereas those of mice treated with CDDP and GA alone are 57.2% and 29.0%, respectively[2]. |
| Cell Assay | The in vitro cell viability effects of Gambogic Acid, CDDP alone, or combined treatments are determined by MTT assay. The cells (2×104 cells per mL) are seeded into 96-well culture plates. After overnight incubation, A549 cells are treated with Gambogic Acid (0.44, 0.88, 1.75, 3.5, 7, 10.5 and 14 μM); NCI-H460 cells are treated with Gambogic Acid (0.5, 1, 2, 4, 8, 12 and 16 μM); NCI-H1299 cells are treated with Gambogic Acid (0.125, 0.25, 0.5, 1, 2 and 4 μM). For the combined treatment in NSCLC cells, three sequences are tested: (a) Gambogic Acid followed by CDDP cells are exposed to Gambogic Acid for 48 h, and then after washout of Gambogic Acid, cells are treated with CDDP for an additional 48 h; (b) CDDP followed by Gambogic Acid cells are exposed to CDDP for 48 h, and then after washout of CDDP, cells are treated with Gambogic Acid for an additional 48 h; and (c) concurrent treatment cells are exposed to both Gambogic Acid and ADM for 48 h. The nature of the drug interaction is analysed by using the combination index (CI)[2]. |
| Animal Admin | Mice[2] To determine the in vivo antitumour activity of Gambogic Acid combined with CDDP, viable A549 cells (5×106/100 μL PBS per mouse) are subcutaneously injected into the right flank of 7- to 8-week-old male SCID mice. When the average tumor volume reach 100 mm3, the mice are randomly divided into four treatment groups, including control (saline only, n=5), Gambogic Acid (3.0 mg/kg per 2 days, intravenously; n=6), CDDP (4 mg/kg per week, intravenously; n=6), and sequential combination (CDDP treatment one day before Gambogic Acid treatment, n=6). CDDP (4 mg/kg, weekly) is generally administered at doses less than the maximum-tolerated dose in an attempt to allow any additive effects of combination treatment with platinum-based agents and Gambogic Acid to be more easily detected. Tumor size is measured once every 2 days with a calipre. Body weight is recorded once every 2 days. After 14 days, the mice are killed and the tumors are excised and stored at -80 °C until further analysis. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 808.9±65.0 °C at 760 mmHg |
| Melting Point | 88.5°C |
| Molecular Formula | C38H44O8 |
| Molecular Weight | 628.751 |
| Flash Point | 251.4±27.8 °C |
| Exact Mass | 628.303589 |
| PSA | 119.36000 |
| LogP | 10.30 |
| Vapour Pressure | 0.0±3.0 mmHg at 25°C |
| Index of Refraction | 1.627 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H315-H319-H335 |
| Precautionary Statements | P261-P301 + P310-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T |
| Risk Phrases | 25-36/37/38 |
| Safety Phrases | 26-45 |
| RIDADR | UN 2811 6.1 / PGIII |
| WGK Germany | 1.0 |
| RTECS | PB9268200 |
| HS Code | 2942000000 |
| HS Code | 2942000000 |
|---|
|
Induction of programmed erythrocyte death by gambogic acid.
Cell Physiol. Biochem. 30(2) , 428-38, (2012) Gambogic acid, a xanthone from Garcinia hanburyi, stimulates apoptosis and has thus anticancer potency. Similar to apoptosis of nucleated cells, erythrocytes may undergo apoptosis-like suicidal death ... |
|
|
Gambogic acid as a non-competitive inhibitor of ATP-binding cassette transporter B1 reverses the multidrug resistance of human epithelial cancers by promoting ATP-binding cassette transporter B1 protein degradation.
Basic Clin Pharmacol Toxicol. 112(1) , 25-33, (2013) Gambogic acid (GA) is known for its anti-cancer activity in a phase II clinical trial. However, the detailed molecular mechanisms of its anti-multidrug resistance remain unclear. The present study was... |
|
|
Subcellular localization and activity of gambogic acid.
ChemBioChem. 13(8) , 1191-8, (2012) The natural product gambogic acid (GA) has shown significant potential as an anticancer agent as it is able to induce apoptosis in multiple tumor cell lines, including multidrug-resistant cell lines, ... |
| GUTTIC ACID |
| (2E)-4-[(2S,17S)-12-Hydroxy-8,21,21-trimethyl-5-(3-methyl-2-buten-1-yl)-8-(4-methyl-3-penten-1-yl)-14,18-dioxo-3,7,20-trioxahexacyclo[15.4.1.0.0.0.0]docosa-4(13),5,9,11,15-pent aen-19-yl]-2-methyl-2-butenoic acid |
| GUTTATIC ACID |
| cambogicacid |
| (2E)-4-[(5S,14aS)-8-hydroxy-3,3,11-trimethyl-13-(3-methylbut-2-en-1-yl)-11-(4-methylpent-3-en-1-yl)-7,15-dioxo-3a,4,5,7-tetrahydro-3H,11H-1,5-methanofuro[3,4-g]pyrano[3,2-b]xanthen-1-yl]-2-methylbut-2-enoic acid |
| 2-Butenoic acid, 2-methyl-4-[(5S,14aS)-3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-buten-1-yl)-11-(4-methyl-3-penten-1-yl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-, (2E)- |
| Gambogic acid |
| (2E)-4-[(2S,17S)-12-Hydroxy-8,21,21-trimethyl-5-(3-methylbut-2-en-1-yl)-8-(4-methylpent-3-en-1-yl)-14,18-dioxo-3,7,20-trioxahexacyclo[15.4.1.0.0.0.0]docosa-4(13),5,9,11,15-pentaen-19-yl]-2-methylbut-2-enoic acid |

