CC-401
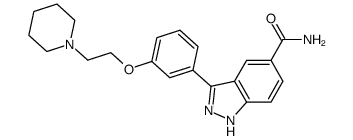
CC-401 structure
|
Common Name | CC-401 | ||
|---|---|---|---|---|
| CAS Number | 395104-30-0 | Molecular Weight | 388.466 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 681.5±65.0 °C at 760 mmHg | |
| Molecular Formula | C22H24N6O | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 366.0±34.3 °C | |
Use of CC-401CC-401 is a potent inhibitor of all three forms of JNK with Ki of 25 to 50 nM. |
| Name | 3-[3-(2-piperidin-1-ylethoxy)phenyl]-5-(1H-1,2,4-triazol-5-yl)-1H-indazole |
|---|---|
| Synonym | More Synonyms |
| Description | CC-401 is a potent inhibitor of all three forms of JNK with Ki of 25 to 50 nM. |
|---|---|
| Related Catalog | |
| Target |
JNK:25-50 nM (Ki) |
| In Vitro | CC-401 has at least 40-fold selectivity for JNK compared with other related kinases, including p38, extracellular signal-regulated kinase (ERK), inhibitor of κB kinase (IKK2), protein kinase C, Lck, zeta-associated protein of 70 kDa (ZAP70). In cell-based assays, 1 to 5 μM CC-401 provides specific JNK inhibition. CC-401, a small molecule that is a specific inhibitor of all three JNK isoforms. CC-401 competitively binds the ATP binding site in JNK, resulting in inhibition of the phosphorylation of the N-terminal activation domain of the transcription factor c-Jun. The specificity of this inhibitor is tested in vitro using osmotic stress of the HK-2 human tubular epithelial cell line. CC-401 inhibits sorbitol-induced phosphorylation of c-Jun in a dosage-dependent manner. However, CC-401 does not prevent sorbitol-induced phosphorylation of JNK, p38, or ERK[1]. |
| In Vivo | The staining of p-JNK is moderately induced in bevazicumab and Oxaliplatin treatments as compared to control, and in the CC-401-treated samples p-cJun content is significantly lower, consistent with effective JNK inhibition. DNA damage is modestly elevated in combined treatments with CC-401[2]. CC-401 treatment from days 7 to 24 slows the progression of proteinuria, which is significantly reduced compared to the no-treatment and vehicle groups at days 14 and 21. However, there is still an increase in the degree of proteinuria at day 21 in CC-401-treated rats compared to proteinuria at day 5. The vehicle and no-treatment groups developed renal impairment at day 24 as shown by an increase in serum creatinine. This is prevented by CC-401 treatment[3]. |
| Cell Assay | Human HK-2 proximal tubular epithelial cells are cultured in DMEM/F12 media supplemented with 10% FCS, 10 ng/mL EGF, and 10 μg/mL bovine pituitary extract. For Western blot studies, cells are seeded into six-well plates and allowed to adhere overnight, and medium is changed to DMEM/F12 supplemented with only 0.5% FCS for 24 h, by which time cells are confluent. CC-401 is prepared in citric acid (pH 5.5) and added to the confluent cells 1 h before the addition of 300 mM sorbitol, and cells are harvested 30 min later using urea-RIPA buffer. Three experiments are performed, each with two replicates per condition. For ELISA experiments, HK-2 cells are seeded into 24-well plates, allowed to adhere overnight, cultured in DMEM/F12 with 0.5% FCS for 24 h, and then incubated with CC-401 or vehicle for 60 min before stimulation with 1 μM Angiotensin II (AngII). Supernatants are harvested 48 h later and assayed for TGF-β1 content using a commercial ELISA kit. Three experiments are performed, each using six replicates per condition[1]. |
| Animal Admin | Mice[2] To assess the efficacy of JNK signaling inhibition by CC-401 in anti-angiogenic and Oxaliplatin combination therapy in a mouse xenograft model, adult (8-10 weeks of age) female severe combined immunodeficient mice (C.B.17 SCID) are used. To generate tumors, HT29 cells (1×106 cells) are injected subcutaneously into the left flank of the mice. When the tumors reached approximately 200 mm3, mice are divided into eight groups (eight mice per group) for treatment with Bevacizumab, Oxaliplatin, CC401, and the appropriate combinations of Bevacizumab, Oxaliplatin and CC-401. Mice in the Bevacizumab treatment group receive 5 mg/kg of Bevacizumab by intraperitoneal injection every 3 days for 21 days. The Oxaliplatin treatment group is injected intraperitoneally with 5 mg/kg Oxaliplatin per week for 2 weeks. The CC-401 treatment group is injected intraperitoneally 25 mg/kg for every 3 days. The combination treatment groups receive Bevacizumab (every 3 days, 5 mg/kg), Oxaliplatin (weekly for 2 weeks, 5 mg/kg), and CC-401 (every 3 days, 25 mg/kg). The control group receive saline intraperitoneally. Tumor volume and body weight are measured every 3 days. Tumor volume is calculated. Tumor growth delay is calculated as the difference in the time for control and treated tumors to grow from 200 to 800 mm3. For tumor growth delay calculations, mice are continued to receive treatments till the tumor volume reached 800 mm3. For immunohistochemistry mice are sacrificed after treatments on day 9 for tumor processing and staining. Rats[3] Female WKY rats (180-220 g) are used. Groups of 9 or 10 rats are immunized by subcutaneous injection of 5 mg of sheep IgG in Freund's complete adjuvant followed 5 days later (termed day 0) by a tail vein injection of sheep anti-rat GBM serum. In this study, CC-401 (200 mg/kg/b.i.d. by oral gavage) or vehicle (sodium citrate) treatment is initiated in groups of 9 or 10 rats at 7 days after anti-GBM serum administration and continued twice daily thereafter until animals are killed at day 24. Additional groups of rats without treatment are killed at day 7 or day 24 after anti-GBM serum injection as controls. Animals are housed in metabolic cages for 22 hours to collect urine on days 5, 14, and 21. Blood is collected at the time of death. Analysis of serum creatinine and urinary protein are performed. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 681.5±65.0 °C at 760 mmHg |
| Molecular Formula | C22H24N6O |
| Molecular Weight | 388.466 |
| Flash Point | 366.0±34.3 °C |
| Exact Mass | 388.201172 |
| PSA | 82.72000 |
| LogP | 3.74 |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.660 |
| Storage condition | 2-8℃ |
|
~90% 
CC-401 CAS#:395104-30-0 |
| Literature: US2006/258706 A1, ; Page/Page column 18; 20 ; |
|
~47% 
CC-401 CAS#:395104-30-0 |
| Literature: US2006/258706 A1, ; Page/Page column 17 ; |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
| 3-(3-(2-(piperidin-1-yl)ethoxy)phenyl)-5-(1H-1,2,4-triazol-3-yl)-1H-indazole |
| CC-401 |
| 3-{3-[2-(1-Piperidinyl)ethoxy]phenyl}-5-(1H-1,2,4-triazol-3-yl)-1H-indazole |
| UNII-NOE38VQA1W |
| RS0046 |
| 1-(5-(1H-1,2,4-triazol-3-yl)(1H-indazol-3-yl))-3-(2-piperidylethoxy)benzene |
| 1-(5-(1H-1,2,4-triazol-5-yl)(1H-indazol-3-yl))-3-(2-piperidylethoxy)benzene |
| 1H-Indazole, 3-[3-[2-(1-piperidinyl)ethoxy]phenyl]-5-(1H-1,2,4-triazol-5-yl)- |