-Trimethoxyflavone
Modify Date: 2024-01-28 12:28:45
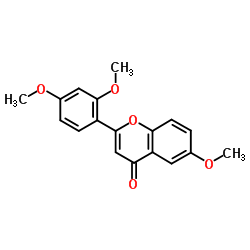
-Trimethoxyflavone structure
|
Common Name | -Trimethoxyflavone | ||
|---|---|---|---|---|
| CAS Number | 720675-74-1 | Molecular Weight | 312.317 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 495.2±45.0 °C at 760 mmHg | |
| Molecular Formula | C18H16O5 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 220.3±28.8 °C | |
Use of -Trimethoxyflavone6,2',4'-Trimethoxyflavone is a potent aryl hydrocarbon receptor (AHR) antagonist. 6,2',4'-Trimethoxyflavone represses AHR-mediated gene induction[1]. |
| Name | 2-(2,4-Dimethoxyphenyl)-6-methoxy-4H-chromen-4-one |
|---|---|
| Synonym | More Synonyms |
| Description | 6,2',4'-Trimethoxyflavone is a potent aryl hydrocarbon receptor (AHR) antagonist. 6,2',4'-Trimethoxyflavone represses AHR-mediated gene induction[1]. |
|---|---|
| Related Catalog | |
| In Vitro | 6,2',4'-trimethoxyflavone (TMF) as an AHR ligand that possesses the characteristics of an antagonist having the capacity to compete with agonists, such as 2,3,7,8-tetrachlorodibenzo-p-dioxin and benzo[a]pyrene, thus effectively inhibiting AHR-mediated transactivation of a heterologous reporter and endogenous targets, e.g., CYP1A1, independent of cell lineage or species. Furthermore, TMF displays superior action by virtue of having no partial agonist activity, in contrast to other documented antagonists, e.g., alpha-napthoflavone, which are partial weak agonists. TMF also exhibits no species or promoter dependence with regard to AHR antagonism[1]. 6,2',4'-Trimethoxyflavone (0-100 μM; 72 hours) shows an inhibitory activity of TNF-⍺ production in THP-1 cells, with IC50 of 2.38 μM. 6,2',4'-Trimethoxyflavone shows an inhibitory activity of TNF-α production in B16-F10 cells with IC50 of 1.32 μM[2]. Cell Viability Assay[2] Cell Line: THP-1 cells, B16-F10 cells Concentration: 0-100 μM Incubation Time: 72 hours Result: Showed inhibitory activity of TNF-⍺ production in THP-1 cells and B16-F10 cells. |
| In Vivo | 6,2',4'-trimethoxyflavone-treated (5 mg/kg/day; i.p.) WT mice shows significantly decreased infarct volume, improved sensorimotor, and nonspatial working memory functions compared with their respective controls[3]. Animal Model: Male C57BL/6 wild-type (WT) mice, AHRcKO mice[3] Dosage: 5 mg/kg/day Administration: I.p. Result: Both TMF-treated and AHRcKO mice attenuated acute cerebral infarction and functional impairments. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 495.2±45.0 °C at 760 mmHg |
| Molecular Formula | C18H16O5 |
| Molecular Weight | 312.317 |
| Flash Point | 220.3±28.8 °C |
| Exact Mass | 312.099762 |
| LogP | 3.54 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.585 |
| 2-(2,4-Dimethoxyphenyl)-6-methoxy-4H-chromen-4-one |
| 4H-1-Benzopyran-4-one, 2-(2,4-dimethoxyphenyl)-6-methoxy- |

