Cefpiramide sodium
Modify Date: 2024-01-02 21:35:21
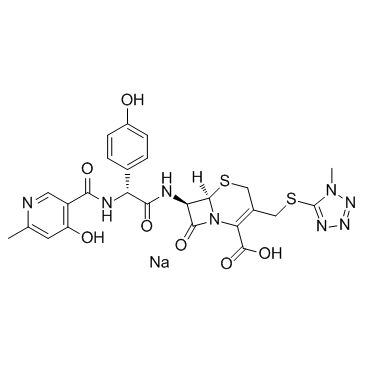
Cefpiramide sodium structure
|
Common Name | Cefpiramide sodium | ||
|---|---|---|---|---|
| CAS Number | 74849-93-7 | Molecular Weight | 635.63 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C25H24N8NaO7S2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of Cefpiramide sodiumCefpiramide sodium (SM-1652; Wy-44635) is a new Pseudomonas-active cephalosporin with a broad spectrum of antibacterial activity.IC50 value:Target: antibacterial agentCefpiramide was moderately susceptible to hydrolysis by a variety of beta-lactamases from Gram-negative bacilli. cefpiramide was more active against Acinetobacter spp. and Pseudomonas spp. Like most other cephalosporins, cefpiramide inhibited methicillin-susceptible staphylococci, non-enterococcal streptococci, Neisseria gonorrhoeae, N. meningitidis and beta-lactamase-negative Haemophilus influenzae [1]. Pharmacokinetic studies in mice showed that cefpiramide attained a peak serum concentration of 12 micrograms/ml and a serum half-life of 40 min, which are higher than attained by cefoperazone with values of 4 micrograms/ml and 18 min. These factors may have caused the combined cefpiramide-gentamicin therapy to result in significantly improved survival rates in mice as well as in higher bactericidal titers than the cefoperazone-gentamicin combination [2].Cefpiramide inhibited many Pseudomonas aeruginosa resistant to carbenicillin, piperacillin, and cefotaxime, but it was less active than ceftazidime and cefsulodin. Cefpiramide inhibited staphylococci and streptococci and had appreciable activity against Streptococcus faecalis and Listeria moncytogenes [3]. |
| Name | cefpiramide sodium |
|---|---|
| Synonym | More Synonyms |
| Description | Cefpiramide sodium (SM-1652; Wy-44635) is a new Pseudomonas-active cephalosporin with a broad spectrum of antibacterial activity.IC50 value:Target: antibacterial agentCefpiramide was moderately susceptible to hydrolysis by a variety of beta-lactamases from Gram-negative bacilli. cefpiramide was more active against Acinetobacter spp. and Pseudomonas spp. Like most other cephalosporins, cefpiramide inhibited methicillin-susceptible staphylococci, non-enterococcal streptococci, Neisseria gonorrhoeae, N. meningitidis and beta-lactamase-negative Haemophilus influenzae [1]. Pharmacokinetic studies in mice showed that cefpiramide attained a peak serum concentration of 12 micrograms/ml and a serum half-life of 40 min, which are higher than attained by cefoperazone with values of 4 micrograms/ml and 18 min. These factors may have caused the combined cefpiramide-gentamicin therapy to result in significantly improved survival rates in mice as well as in higher bactericidal titers than the cefoperazone-gentamicin combination [2].Cefpiramide inhibited many Pseudomonas aeruginosa resistant to carbenicillin, piperacillin, and cefotaxime, but it was less active than ceftazidime and cefsulodin. Cefpiramide inhibited staphylococci and streptococci and had appreciable activity against Streptococcus faecalis and Listeria moncytogenes [3]. |
|---|---|
| Related Catalog | |
| References |
| Molecular Formula | C25H24N8NaO7S2 |
|---|---|
| Molecular Weight | 635.63 |
| PSA | 266.19000 |
| Storage condition | -20℃ |
| Cefpiramide sodium |
| SM 1652 |
| Suncefal |
| Cefpriamide free acid |
| Antibiotic SM 1652 |
| 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,7-[[(2R)-[[(4-hydroxy-6-methyl-3-pyridinyl)carbonyl]amino](4-hydroxyphenyl)acetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-,monosodium salt,(6R,7R) |
| Cefpiran |
| Sodium (6R,7R)-7-[[(2R)-2-(4-hydroxyphenyl)-2-[(6-methyl-4-oxo-1H-pyridine-3-carbonyl)amino]acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate |
| Sepatren |