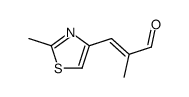
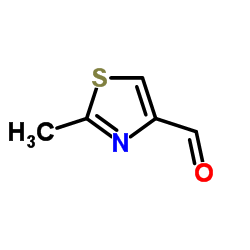
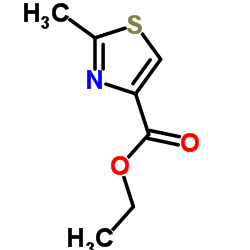
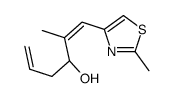
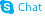152044-53-6
| Name | (1R,5S,6S,7R,10S,14S,16S)-6,10-dihydroxy-5,7,9,9-tetramethyl-14-[(E)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-13,17-dioxabicyclo[14.1.0]heptadecane-8,12-dione |
|---|---|
| Synonyms |
UNII-51E07YBX96
Epothilone A (-)-Epothilone A Epo A 4,17-Dioxabicyclo[14.1.0]heptadecane-5,9-dione, 7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(E)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-, (1S,3S,7S,10R,11S,12S,16R)- [1R*,3R*(E),7R*,10S*,11R*,12R*,16S*]-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione Epothilon A (1S,3S,7S,10R,11S,12S,16R)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(1E)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione (1S,3S,7S,10R,11S,12S,16R)-7,11-Dihydroxy-8,8,10,12-tetramethyl-3-[(1E)-1-(2-methyl-1,3-thiazol-4-yl)-1-propen-2-yl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione 4,17-Dioxabicyclo[14.1.0]heptadecane-5,9-dione, 7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(E)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-, (1R,3R,7R,10S,11R,12R,16S)- (1R,3R,7R,10S,11R,12R,16S)-7,11-Dihydroxy-8,8,10,12-tetramethyl-3-[(1E)-1-(2-methyl-1,3-thiazol-4-yl)-1-propen-2-yl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione epithilone A Epothilones (1S,3S,7S,10R,11S,12S,16R)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(1E)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione |
| Description | Epothilone A is a competitive inhibitor of the binding of [3H] paclitaxel to tubulin polymers, with a Ki of 0.6-1.4 μM. |
|---|---|
| Related Catalog | |
| In Vitro | Epothilone A is a competitive inhibitor of the binding of [3H] paclitaxel to tubulin polymers. The apparent Ki value for Epothilone A is 1.4 μM by Hanes analysis and 0.6 μM by Dixon analysis[1]. Epothilone A, is noted to be highly cytotoxic (IC50=0.05 μM) in vitro when applied to the human T-24 bladder carcinoma cell line. The binding affinity of Epothilone A to tubulin is of the same order of magnitude as the binding affinity of paclitaxel to tubulin based on competition assays. The IC50 for displacement of 100 nM of (3H) paclitaxel from the tubulin binding site is 3.6 μM for paclitaxel, 2.3 μM for Epothilone A, and 3.3 μM for patupilone[2]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 683.3±55.0 °C at 760 mmHg |
| Melting Point | 95ºC |
| Molecular Formula | C26H39NO6S |
| Molecular Weight | 493.656 |
| Flash Point | 367.1±31.5 °C |
| Exact Mass | 493.249817 |
| PSA | 137.49000 |
| LogP | 2.54 |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.532 |
| Storage condition | -20°C |
| RIDADR | NONH for all modes of transport |
|---|
| Precursor 7 | |
|---|---|
| DownStream 1 | |