168626-94-6
| Name | conivaptan hydrochloride |
|---|---|
| Synonyms |
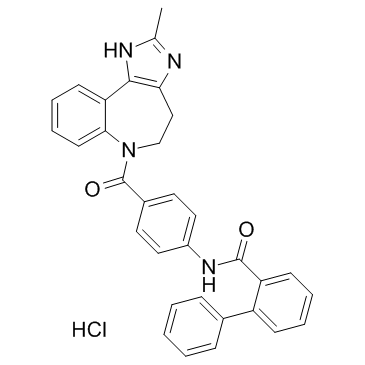
(1,1'-Biphenyl)-2-carboxamide,N-(4-((4,5-dihydro-2-methylimidazo(4,5-d)(1)benzazepin-6(1H)-yl)carbonyl)phenyl)-,monohydrochloride
Conivaptan hydrochloride N-{4-[(2-methyl-4,5-dihydroimidazo[4,5-d][1]benzazepin-6(1H)-yl)carbonyl]phenyl}biphenyl-2-carboxamide hydrochloride YM-087 UNII-75L57R6X36 N-[4-(2-methyl-4,5-dihydro-3H-imidazo[4,5-d][1]benzazepine-6-carbonyl)phenyl]-2-phenylbenzamide,hydrochloride [1,1'-Biphenyl]-2-carboxamide, N-[4-[(4,5-dihydro-2-methylimidazo[4,5-d][1]benzazepin-6(1H)-yl)carbonyl]phenyl]-, hydrochloride (1:1) N-{4-[(2-Methyl-4,5-dihydroimidazo[4,5-d][1]benzazepin-6(1H)-yl)carbonyl]phenyl}-2-biphenylcarboxamide hydrochloride (1:1) Vaprisol CI-1025 Conivaptanhydrochloride [1,1'-biphenyl]-2-carboxamide, N-[4-[(4,5-dihydro-2-methylimidazo[4,5-d][1]benzazepin-6(1H)-yl)carbonyl]phenyl]-, monohydrochloride Conivaptan HCl Conivaptan hydrochloride [USAN] Conivaptan HCI Conivaptan (hydrochloride) |
| Description | Conivaptan (hydrochloride) is a non-peptide antagonist of vasopressin receptor, with Ki values of 0.48 and 3.04 nM for rat liver V1A receptor and rat kidney V2 receptor respectively. |
|---|---|
| Related Catalog | |
| Target |
Ki: 0.48 nM (V1A receptor), 3.04 nM (V2 receptor) |
| In Vivo | Conivaptan (0.03, 0.1 and 0.3 mg/kg, i.v.) dose-dependently increases urine volume and reduces urine osmolality in both myocardial infarction and sham-operated rats. Conivaptan (0.3 mg/kg i.v.) significantly reduces right ventricular systolic pressure, left ventricular end-diastolic pressure, lung/body weight and right atrial pressure in myocardial infarction rats. Conivaptan (0.3 mg/kg i.v.) significantly increases dP/dt(max)/left ventricular pressure in myocardial infarction rats[1]. Conivaptan produces an acute increase in urine volume (UV), a reduction in osmolality (UOsm) and, at the end of the investigation, cirrhotic rats receiving the V(1a)/V(2)-AVP receptor antagonist does not show hyponatremia or hypoosmolality. Conivaptan also normalizes U(Na)V without affecting creatinine clearance and arterial pressure[2]. Conivaptan (0.01 to 0.1 mg/kg, i.v.) exerts a dose-dependent diuretic effect in dogs without an increase in the urinary excretion of electrolytes, inhibits the pressor effect of exogenous vasopressin in a dose-dependent manner (0.003 to 0.1 mg/kg i.v.) and, at the highest dose (0.1 mg/kg i.v.), almost completely blocks vasoconstriction caused by exogenous vasopressin. Conivaptan (0.1 mg/kg, i.v.) improves cardiac function, as evidenced by significant increases in left ventricular dP/dtmax, cardiac output and stroke volume, and reduces preload and afterload, as evidenced by significant decreases in left ventricular end-diastolic pressure and total peripheral vascular resistance in dogs with congestive heart failure[3]. |
| Animal Admin | At 4 weeks after the operation, 39 myocardial infarction rats survived. Thirty are randomly selected without bias and divided into five groups such that the distribution of infarct size and body weight among groups are similar, and given vehicle, conivaptan (0.03, 0.1 and 0.3 mg/kg) or SR121463A (0.3 mg/kg) by intravenous administration. Sham rats are also divided into four groups and given vehicle or conivaptan (0.03, 0.1 and 0.3 mg/kg) by intravenous administration. Rats are then placed individually in metabolic cages and urine is collected for 3 h. Urine osmolality is measured by the freezing point depression method using an osmometer. |
| References |
| Boiling Point | 751.2ºC at 760 mmHg |
|---|---|
| Melting Point | >250° |
| Molecular Formula | C32H27ClN4O2 |
| Molecular Weight | 535.035 |
| Flash Point | 408.1ºC |
| Exact Mass | 534.182251 |
| PSA | 78.09000 |
| LogP | 7.44730 |
| Vapour Pressure | 1.89E-22mmHg at 25°C |
| Storage condition | -20°C |
| Hazard Codes | Xi |
|---|