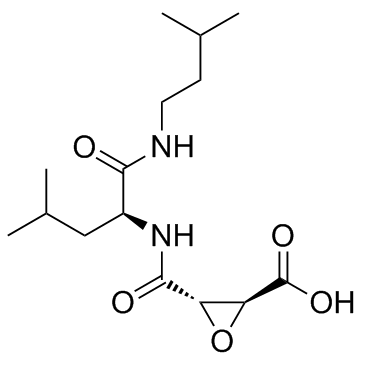
76684-89-4
| Name | e-64c |
|---|---|
| Synonyms |
2-Oxiranecarboxylic acid, 3-[[[(1S)-3-methyl-1-[[(3-methylbutyl)amino]carbonyl]butyl]amino]carbonyl]-, (2S,3S)-
Loxistatin acid (2S,3S)-3-({(2S)-4-Methyl-1-[(3-methylbutyl)amino]-1-oxo-2-pentanyl}carbamoyl)-2-oxiranecarboxylic acid (2S,3S)-3-({(2S)-4-methyl-1-[(3-methylbutyl)amino]-1-oxopentan-2-yl}carbamoyl)oxirane-2-carboxylic acid L-TRANS-EPOXYSUCCINYL-LEU-3-METHYLBUTYLAMIDE E-64c,EP 475 Inhibitorforthiolprotease anecarboxylicacid |
| Description | E 64c is a derivative of naturally occurring epoxide inhibitor of cysteine proteases, a Calcium-activated neutral protease (CANP) inhibitor and a very weak irreversible cathepsin C inhibitor. |
|---|---|
| Related Catalog | |
| Target |
Cysteine proteases[1], CANP[2], Cathepsin C[3]. |
| In Vitro | E-64c, a derivative of naturally occurring epoxide inhibitor of cysteine proteases, with papain; especially with regard to the hydrogen bonding and hydrophobic interactions of the ligands with conserved residues in the catalytic binding site[1]. E 64c (k2/Ki=140±5M-1s-1) is demonstrated to be a lead structure for the development of irreversible cathepsin C inhibitors[3]. |
| In Vivo | The t-1/2 of plasma E-64c is 0.48 hours. The hemodynamic effects of E-64c are absent at this dose. Using two way analysis of variance, the effects of reperfusion (p=0.0016) or E-64c (p=0.0226) per se on infarct size are significant. In comparing Group A with Group B and Group C with Group D, the depletion of CPK in the E-64c treated groups (Groups A and C) is slightly less than in the vehicle-injected groups (Groups B and D). The insufficient effect of E-64c alone may be explained by the early administration and relatively short t-1/2. Since the effectiveness of NCO-700 has been established,6),7) our findings might indicate a small but beneficial effect of E-64c on infarct size and CPK content[2]. |
| Animal Admin | Dogs[2] Studies are carried out in 83 mongrel dogs with a mean weight of 11.2kg. They are anesthetized with intravenous sodium thiamylal (7mg/kg). An intravenous bolus of E-64c (100mg/kg), dissolved in saturated sodium bicarbonate, is administered immediately before the occlusion and after reperfusion in Group A (n=17), whereas Group B (n=17) receive only the vehicle solution at these times. In the remaining 49 dogs (Groups C and D), the LAD is permanently ligated at the same level and an intravenous bolus of either Loxistatin acid (100mg/kg) (Group C; n=24) or vehicle only (Group D; n=25) is given immediately before and 1 hour after the ligation. The dose of E-64c is designed for its possible use in clinical practice and the estimated intramyocardial Loxistatin acid molecular concentration is 1,000 times that of total mCANP[2]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 596.4±50.0 °C at 760 mmHg |
| Molecular Formula | C15H26N2O5 |
| Molecular Weight | 314.377 |
| Flash Point | 314.5±30.1 °C |
| Exact Mass | 314.184174 |
| PSA | 108.03000 |
| LogP | 1.36 |
| Vapour Pressure | 0.0±3.6 mmHg at 25°C |
| Index of Refraction | 1.504 |
| Storage condition | −20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | RR0404200 |