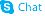1187594-09-7
| Name | 2-[1-ethylsulfonyl-3-[4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)pyrazol-1-yl]azetidin-3-yl]acetonitrile |
|---|---|
| Synonyms |
INCB028050
2-(3-(4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl)-1-(ethylsulfonyl)azetidin-3-yl)acetonitrile {1-(Ethylsulfonyl)-3-[4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl]-3-azetidinyl}acetonitrile {1-(ethylsulfonyl)-3-[4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl]azetidin-3-yl}ethanenitrile 3-Azetidineacetonitrile, 1-(ethylsulfonyl)-3-[4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl]- LY3009104 {1-(ethylsulfonyl)-3-[4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl]azetidin-3-yl}acetonitrile Baricitinib 3-Azetidineacetonitrile,1-(ethylsulfonyl)-3-(4-(7H-pyrrolo(2,3-d)pyrimidin-4-yl)-1H-pyrazol-1-yl) |
| Description | Baricitinib is a selective and orally bioavailable JAK1 and JAK2 inhibitor with IC50s of 5.9 nM and 5.7 nM, respectively. |
|---|---|
| Related Catalog | |
| Target |
JAK2:5.7 nM (IC50) JAK1:5.9 nM (IC50) Tyk2:53 nM (IC50) JAK3:560 nM (IC50) |
| In Vitro | In cell-based assays, Baricitinib (INCB028050) proves to be a potent inhibitor of JAK signaling and function. In PBMCs, Baricitinib inhibits IL-6-stimulated phosphorylation of the canonical substrate STAT3 (pSTAT3) and subsequent production of the chemokine MCP-1 with IC50 values of 44 nM and 40 nM, respectively. In isolated naive T-cells, INCB028050 also inhibits pSTAT3 stimulated by IL-23 (IC50=20 nM). Importantly, this inhibition prevented the production of two pathogenic cytokines (IL-17 and IL-22) produced by Th17 cells-a subtype of helper T cells with demonstrable inflammatory and pathogenic properties-with an IC50 value of 50 nM. In stark contrast, the structurally similar but ineffective JAK1/2 inhibitors INCB027753 and INCB029843 has no significant effect in any of these assays systems when tested at concentrations up to 10 μM[1]. |
| In Vivo | Baricitinib (INCB028050) treatment, compares with vehicle, inhibits the increase in hind paw volumes during the 2 wk of treatment by 50% at a dose of 1 mg/kg and >95% at doses of 3 or 10 mg/kg. Because baseline paw volume measurements are taken on treatment day 0-in animals with significant signs of disease-it is possible to have >100% inhibition in animals showing marked improvement in swelling[1]. Baricitinib (0.7 mg/day) treated mice exhibits substantially reduced inflammation as assessed by H&E staining, reduced CD8 infiltration, and reduced MHC class I and class II expression when compared with vehicle-control treated mice. CD8+NKG2D+ cells, critical effectors of disease in murine and human alopecia areata (AA), are greatly diminished in Baricitinib treated mice compare with vehicle control treated mice[2]. |
| Kinase Assay | Enzyme assays are performed using a homogeneous time-resolved fluorescence assay with recombinant epitope tagged kinase domains (JAK1, 837-1142; JAK2, 828-1132; JAK3, 718-1124; Tyk2, 873-1187) or full-length enzyme (cMET and Chk2) and peptide substrate. Each enzyme reaction is performed with or without test compound (11-point dilution), JAK, cMET, or Chk2 enzyme, 500 nM (100 nM for Chk2) peptide, ATP (at the Km specific for each kinase or 1 mM), and 2.0% DMSO in assay buffer. The calculated IC50 value is the compound concentration required for inhibition of 50% of the fluorescent signal. Additional kinase assays are performed at Cerep using standard conditions at 200 nM. Enzymes tested included: Abl, Akt1, AurA, AurB, CDC2, CDK2, CDK4, CHK2, c-kit, EGFR, EphB4, ERK1, ERK2, FLT-1, HER2, IGF1R, IKKα, IKKβ, JNK1, Lck, MEK1, p38α, p70S6K, PKA, PKCα, Src, and ZAP70[1]. |
| Cell Assay | Human PBMCs are isolated by leukapheresis followed by Ficoll-Hypaque centrifugation. For the determination of IL-6-induced MCP-1 production, PBMCs are plated at 3.3×105 cells per well in RPMI 1640+10% FCS in the presence or absence of various concentrations of INCB028050 (1 nM, 10 nM, 100 nM, 1 μM, and 10 μM). Following preincubation with compound for 10 min at room temperature, cells are stimulated by adding 10 ng/mL human recombinant IL-6 to each well. Cells are incubated for 48 h at 37°C, 5% CO2. Supernatants are harvested and analyzed by ELISA for levels of human MCP-1. The ability of INCB028050 to inhibit IL-6-induced secretion of MCP-1 is reported as the concentration required for 50% inhibition (IC50). Proliferation of Ba/F3-TEL-JAK3 cells is performed over 3 d using Cell-Titer Glo[1]. |
| Animal Admin | Rats[1] Female rats (n=6 per gender per group) are given a dose of 10 mg/kg Baricitinib and given by oral gavage at 10 mL/kg. The first three rats are bled at 0 (predose), 2, 8, and 24 h, and the second three rats are bled 1, 4, and 12 h after dosing. EDTA is used as the anticoagulant, and samples are centrifuged to obtain plasma. An analytical method for the quantification of INCB028050 has been developed and used to analyze samples from toxicology studies. The method combines a protein precipitation extraction with 10% methanol in acetonitrile and LC/MS/MS analysis. The method has demonstrated a linear assay range 1-5000 nM using 0.1 mL of study samples. Data are processed using Analyst 1.3.1. A standard curve is determined from peak area ratio versus concentration using a weighted linear regression (1/x2). Mice[2] The C3H/HeJ graft-recipient mouse model of AA is used for these experiments. Briefly, alopecic skin from a C3H/HeJ mouse that spontaneously developed hair loss is grafted onto 8-10 week old C3H/HeJ mice free of disease. At the time of grafting, an osmotic pump that administered approximately 0.7 mg/day of Baricitinib or placebo is implanted. Osmotic pumps are changed monthly. A time-to-event survival analysis for interval censored data is performed. The survival and interval packages in R are used to perform log-rank tests. The hypothesis that the survival distributions are equal in the (n=10) Baricitinib-treated mice and (n=10) placebo-treated mice is rejected at the 5% level using Sun's score to perform an exact log-rank two-sample test with the p-value of 0.0035. |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 707.2±70.0 °C at 760 mmHg |
| Molecular Formula | C16H17N7O2S |
| Molecular Weight | 371.417 |
| Flash Point | 381.5±35.7 °C |
| Exact Mass | 371.116455 |
| PSA | 128.94000 |
| LogP | -0.06 |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.763 |
| Hazard Codes | Xi |
|---|
|
~86% 
1187594-09-7 |
| Literature: INCYTE CORPORATION Patent: US2009/233903 A1, 2009 ; Location in patent: Page/Page column 69 ; US 20090233903 A1 |
|
~85% 
1187594-09-7 |
| Literature: INCYTE CORPORATION Patent: US2009/233903 A1, 2009 ; Location in patent: Page/Page column 58; 60 ; US 20090233903 A1 |
| Precursor 0 | |
|---|---|
| DownStream 1 | |