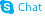5-Iodouridine
5-Iodouridine structure
|
Common Name | 5-Iodouridine | ||
|---|---|---|---|---|
| CAS Number | 1024-99-3 | Molecular Weight | 370.10 | |
| Density | 2.3±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C9H11IN2O6 | Melting Point | 205-207ºC (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
Use of 5-Iodouridine5-Iodouridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc[1]. |
| Name | 5-Iodouridine |
|---|---|
| Synonym | More Synonyms |
| Description | 5-Iodouridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 2.3±0.1 g/cm3 |
|---|---|
| Melting Point | 205-207ºC (dec.)(lit.) |
| Molecular Formula | C9H11IN2O6 |
| Molecular Weight | 370.10 |
| Exact Mass | 369.966156 |
| PSA | 124.78000 |
| LogP | -0.43 |
| Index of Refraction | 1.748 |
| Storage condition | 2-8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 22-24/25-36/37/39-27-26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2934999090 |
|
~92% 
5-Iodouridine CAS#:1024-99-3 |
| Literature: Kumar; Wiebe; Knaus Canadian Journal of Chemistry, 1994 , vol. 72, # 9 p. 2005 - 2010 |
|
~94% 
5-Iodouridine CAS#:1024-99-3 |
| Literature: Asakura, Jun-ichi; Robins, Morris J. Journal of Organic Chemistry, 1990 , vol. 55, # 16 p. 4928 - 4933 |
|
~% 
5-Iodouridine CAS#:1024-99-3 |
| Literature: Journal of Organic Chemistry, , vol. 55, # 16 p. 4928 - 4933 |
|
~% 
5-Iodouridine CAS#:1024-99-3 |
| Literature: Angewandte Chemie - International Edition, , vol. 50, # 9 p. 2155 - 2158 |
| Precursor 3 | |
|---|---|
| DownStream 10 | |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Loss of Foxm1 Results in Reduced Somatotrope Cell Number during Mouse Embryogenesis.
PLoS ONE 10 , e0128942, (2015) FOXM1, a member of the forkhead box transcription factor family, plays a key role in cell cycling progression by regulating the expression of critical G1/S and G2/M phase transition genes. In vivo stu... |
|
|
Synthesis of uridine phosphoramidite analogs: reagents for site-specific incorporation of photoreactive sites into RNA sequences.
Bioconjug. Chem. 5(6) , 508-12, (1994) The synthesis of three new photoactive RNA phosphoramidites, 5-bromouridine, 5-iodouridine, and O4-triazolouridine, is reported. The 5' OH of bromouridine and iodouridine were protected as dimethoxytr... |
|
|
Phosphine containing oligonucleotides for the development of metallodeoxyribozymes.
Chem. Commun. (Camb.) (15) , 1556-8, (2007) Novel transition metal catalysts based on oligonucleotides can be easily obtained by functionalization of 5-iodouridine with phosphine ligands, resulting in good asymmetric induction in palladium cata... |
| 5-Iodouridine |
| 5-Iod-uridin |
| 5-Iodoruidine |
| 2,4-Dihydroxy-5-iodo-1-β-D-ribofuranosylpyrimidine |
| Uridine, 5-iodo- |
| 5-iodo-uridine |
| EINECS 213-833-1 |
| 5-Jod-uridin |
| 5-Iodo-D-uridine |
| MFCD00006532 |

 CAS#:59240-49-2
CAS#:59240-49-2![5-[3-(Trifluoroacetamido)-1-(E)-propenyl]-uridine structure](https://www.chemsrc.com/caspic/131/869222-68-4.png) CAS#:869222-68-4
CAS#:869222-68-4 CAS#:18646-11-2
CAS#:18646-11-2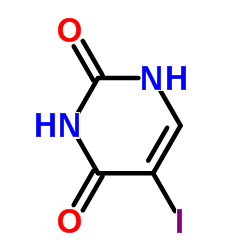 CAS#:696-07-1
CAS#:696-07-1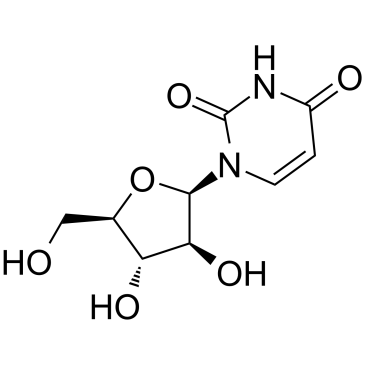 CAS#:3083-77-0
CAS#:3083-77-0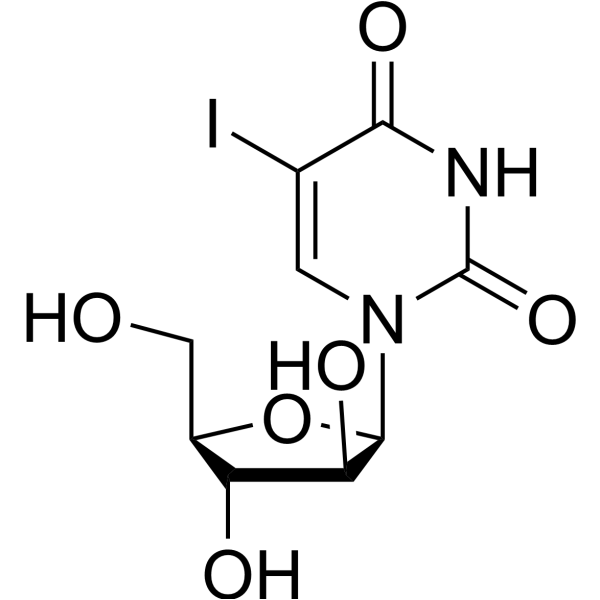 CAS#:3052-06-0
CAS#:3052-06-0![1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-(2-phenylethynyl)pyrimidine-2,4-dione structure](https://www.chemsrc.com/caspic/168/649558-84-9.png) CAS#:649558-84-9
CAS#:649558-84-9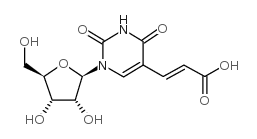 CAS#:99394-52-2
CAS#:99394-52-2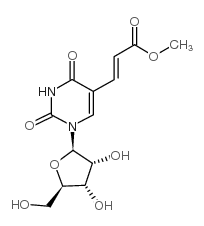 CAS#:58931-19-4
CAS#:58931-19-4