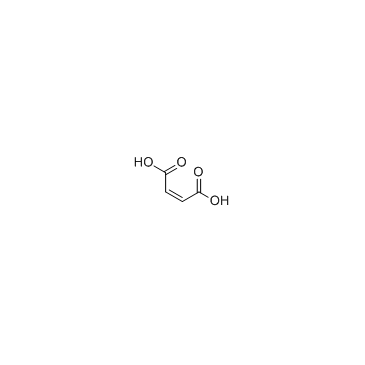
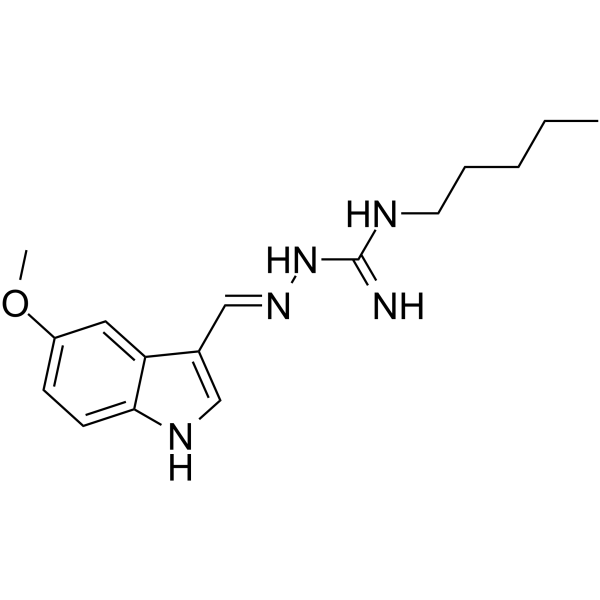
Tegaserod maleate
Modify Date: 2024-01-02 16:39:56
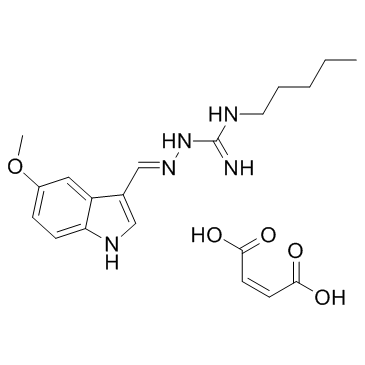
Tegaserod maleate structure
|
Common Name | Tegaserod maleate | ||
|---|---|---|---|---|
| CAS Number | 189188-57-6 | Molecular Weight | 417.459 | |
| Density | N/A | Boiling Point | 661.4ºC at 760 mmHg | |
| Molecular Formula | C20H27N5O5 | Melting Point | 180-183ºC | |
| MSDS | Chinese USA | Flash Point | 353.8ºC | |
Use of Tegaserod maleateTegaserod maleate is a partial agonist of the 5-HT4 receptor; stimulates the peristaltic reflex and accelerates gastrointestinal transit.IC50 value:Target: 5-HT4 agonistIn an in vivo model for peripheral nerve regeneration, mice receiving tegaserod at the site of injury showed enhanced recovery compared to control mice receiving vehicle control as evidenced by functional measurements and histology [1]. Treatment with fluoxetine (10 mg · kg(-1) · day(-1), days 36-42), tegaserod (1 mg · kg(-1) · day(-1), day 43), or the combination of both, reduced visceral hypersensitivity and plasma 5-HT levels [2]. Intravenous or intraduodenal tegaserod (0.3-1.0 mg.kg(-1)) had no inhibitory effect on mesenteric and colonic blood flow. Peroral treatment of rats with alosetron or tegaserod for 7 days did not modify mesenteric haemodynamics at baseline and after blockade of nitric oxide synthesis [3]. |
| Name | tegaserod maleate |
|---|---|
| Synonym | More Synonyms |
| Description | Tegaserod maleate is a partial agonist of the 5-HT4 receptor; stimulates the peristaltic reflex and accelerates gastrointestinal transit.IC50 value:Target: 5-HT4 agonistIn an in vivo model for peripheral nerve regeneration, mice receiving tegaserod at the site of injury showed enhanced recovery compared to control mice receiving vehicle control as evidenced by functional measurements and histology [1]. Treatment with fluoxetine (10 mg · kg(-1) · day(-1), days 36-42), tegaserod (1 mg · kg(-1) · day(-1), day 43), or the combination of both, reduced visceral hypersensitivity and plasma 5-HT levels [2]. Intravenous or intraduodenal tegaserod (0.3-1.0 mg.kg(-1)) had no inhibitory effect on mesenteric and colonic blood flow. Peroral treatment of rats with alosetron or tegaserod for 7 days did not modify mesenteric haemodynamics at baseline and after blockade of nitric oxide synthesis [3]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 661.4ºC at 760 mmHg |
|---|---|
| Melting Point | 180-183ºC |
| Molecular Formula | C20H27N5O5 |
| Molecular Weight | 417.459 |
| Flash Point | 353.8ºC |
| Exact Mass | 417.201233 |
| PSA | 149.45000 |
| LogP | 3.10190 |
| Vapour Pressure | 2.15E-18mmHg at 25°C |
| Storage condition | -20°C Freezer |
| RIDADR | NONH for all modes of transport |
|---|---|
| HS Code | 2933990090 |
|
~72% 
Tegaserod maleate CAS#:189188-57-6 |
| Literature: Venkataraman, Sundaram; Gudipati, Srinivasulu; Mandava Venkata Naga, Brahmeshwararao; Banda, Goverdhan Reddy; Singamsetty, Radhakrishna Patent: US2005/272802 A1, 2005 ; Location in patent: Page/Page column 2-3 ; |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
| (2E)-2-[(5-Methoxy-1H-indol-3-yl)methylene]-N-pentylhydrazinecarboximidamide (2Z)-2-butenedioate |
| Zelnorm |
| Hydrazinecarboximidamide, 2-[(5-methoxy-1H-indol-3-yl)methylene]-N-pentyl-, (2E)-, (2Z)-2-butenedioate (1:1) |
| (2E)-2-[(5-methoxy-1H-indol-3-yl)methylidene]-N-pentylhydrazinecarboximidamide (2Z)-but-2-enedioate (1:1) |
| Zelmac |
| MFCD04116201 |
| acide (2Z)-but-2-ènedioïque - (2E)-2-[(5-méthoxy-1H-indol-3-yl)méthylidène]-N-pentylhydrazinecarboximidamide (1:1) |
| Tegaserod Maleate |
| (2E)-2-[(5-Methoxy-1H-indol-3-yl)methylene]-N-pentylhydrazinecarboximidamide (2Z)-but-2-enedioate (1:1) |
| (2Z)-But-2-endisäure--(2E)-2-[(5-methoxy-1H-indol-3-yl)methyliden]-N-pentylhydrazincarboximidamid(1:1) |
| (2E)-2-[(5-Methoxy-1H-indol-3-yl)methylene]-N-pentylhydrazinecarboximidamide (2Z)-2-butenedioate (1:1) |
| (2E)-2-[(5-methoxy-1H-indol-3-yl)methylidene]-N-pentylhydrazinecarboximidamide (2Z)-but-2-enedioate |
| Tegaserod (maleate) |