34367-04-9
| Name | GinsenosideRo |
|---|---|
| Synonyms |
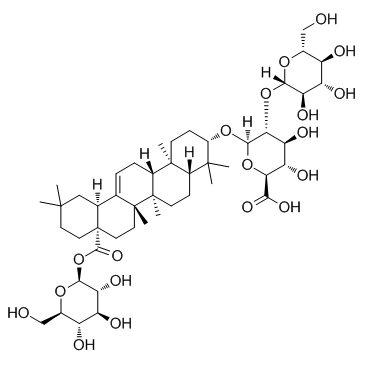
β-D-Glucopyranose, 1-O-[(3β)-3-[(2-O-β-D-glucopyranosyl-β-D-glucopyranuronosyl)oxy]-28-oxoolean-12-en-28-yl]-
glycoside L2 HericiuMsaponin S3 chikusetsusaponin V Chikusetusaponin V Ginsenoside Ro PolysciasaponinP3 1-O-[(3β)-3-{[2-O-(β-D-Glucopyranosyl)-β-D-glucopyranuronosyl]oxy}-28-oxoolean-12-en-28-yl]-β-D-glucopyranose Chikusetsusaponin 5 chikusetsusaponin-V ginsenoside-R0 |
| Description | Ginsenoside Ro exhibits a Ca2+-antagonistic antiplatelet effect with an IC50 of 155 μM. Ginsenoside Ro reduces the production of TXA2 more than it reduces the activities of COX-1 and TXAS. |
|---|---|
| Related Catalog | |
| Target |
Ca2+ TXA2 |
| In Vitro | Ginsenoside Ro in Panax ginseng is a beneficial novel Ca2+-antagonistic compound and may prevent platelet aggregation-mediated thrombotic disease. Ginsenoside Ro dose-dependently reduces thrombin-stimulated platelet aggregation, and IC50 is approximately 155 μM[1]. Ginsenoside Ro inhibits TXA2 production to abolish thrombin-induced platelet aggregation. Thromboxane A2 (TXA2) induces platelet aggregation and promotes thrombus formation. Ginsenoside Ro dose-dependently (50-300 μM) reduces the TXB2 level that is induced by thrombin; Ginsenoside Ro (300 μM) inhibits the thrombin-mediated elevation in TXB2 level by 94.9%. COX-1 activity in the absence of Ginsenoside Ro (negative control) is 2.3±0.1 nmol/mg protein. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, COX-1 activity is reduced by 26.4% of that of the negative control. TXA2 synthase (TXAS) activity in the absence of Ginsenoside Ro (negative control) is 220.8±1.8 ng/mg protein/min. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, TXAS activity is reduced by 22.9% of that of the negative control. The inhibitory effect of Ginsenoside Ro (300 μM) on TXB2 production (94.9%) is significantly higher than those on COX-1 (26.4%) and TXAS (22.9%) activities[2]. To assess the toxicity of Ginsenoside Ro in Raw 264.7 cells, they are first treated with various concentrations (10 μM, 50 μM, 100 μM, and 200 μM) of Ginsenoside Ro for 24 h. Ginsenoside Ro exhibits no significant dose dependent toxicity. The effect of Ginsenoside Ro is next determined on cell viability and ROS levels, a marker of oxidative stress, following treatment with 1 μg/mL LPS. LPS reduces cell viability by ∼70% compared with nontreated controls. Pretreatment with 100 μM and 200 μM Ginsenoside Ro for 1 h prior to 1 μg/mL LPS incubation for 24 h leads to a significant increase in cell viability. The changes in ROS levels and NO production are consistent with the effects of Ginsenoside Ro on viability[3]. |
| In Vivo | Ginsenoside Ro dissolved in water is administrated by gavage to mice at doses of 25 and 250 mg/kg/day for 4 days before i.v. injection of HT29 in order to keep blood concentrations of Ginsenoside Ro above a certain level before HT29 i.v. injection followed by 40 days of oral administration of Ginsenoside Ro to the mice. After 38 days of treatment, the animals are euthanized, and the number of pulmonary metastatic nodules is counted in addition to evaluation of toxicity of Ginsenoside Ro and mouse pathology by HT29. Ginsenoside Ro (250 mg/kg/day) produces a significant decrease in the number of tumor nodules on the lung surface, yielding inhibition rates of 88% (P < 0.01)[4]. |
| Kinase Assay | The microsomal fraction of platelets is preincubated with Ozagrel (11 nM, IC50), a positive control, or with various concentrations of Ginsenoside Ro and other reagents at 37°C for 5 min. The reaction is initiated by adding prostaglandin H2, and the samples are incubated at 37°C for 1 min; the reaction is terminated by adding citric acid (1 M). After neutralization with 1 N NaOH, the amount of TXB2 is determined using a TXB2 EIA kit[2]. |
| Cell Assay | Cell viability is determined with an MTT assay kit. Briefly, Raw 264.7 cells are plated in 48-well plates at a density of 2.0×104 cells per well, incubated for 24 h, and treated with various concentrations of Ginsenoside Ro for 24 h. How 1 h of pretreatment with Ginsenoside Ro (50 μM, 100 μM, and 200 μM) affects the viability of Raw 264.7 cells is then investigated treated with 1 μg/mL LPS for 24 h. After the incubation period, 10 μL of MTT reagent is added to each well and incubated for 3 h at 37°C in 5% CO2. The resulting formazan crystals are subsequently dissolved in MTT solubilization solution. The absorbance is determined at 540 nm using a microplate reader[3]. |
| Animal Admin | Mice[4] Female BALB/c mice (20-25 g, 6-8 weeks old) are used. The experimental model of lung metastasis is established by tail vein injection of HT29 cells to mimic the dissemination of CTCs. HT29 cells in the number of 2×106 cells in 0.2 mL PBS are infected into the tail vein of six-week-old female Balb/c mice. Before the HT29 inoculation, oral gavage pretreatment of PBS-suspended B (Ginsenoside Ro) is given daily for 4 days, followed by a 40-day treatment. Treatment groups (N = 10) include: 0 mg/kg, 25 mg/kg and 250 mg/kg Ginsenoside Ro. Body weight is measured and recorded every four days. Mice are sacrificed after 40 days of tumor metastasis and growth and 44 days of treatment with B. The number of surface lung metastasis nodules is evaluated in each treatment group. Slides with 4-5 μm thick lung section are prepared, paraffin embedded and then stained with hematoxylin and eosin[4]. |
| References |
| Density | 1.14 |
|---|---|
| Boiling Point | 1018.6±65.0 °C at 760 mmHg |
| Melting Point | 241 °C |
| Molecular Formula | C48H76O19 |
| Molecular Weight | 957.106 |
| Flash Point | 289.2±27.8 °C |
| Exact Mass | 956.498108 |
| PSA | 312.05000 |
| LogP | 6.29 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.627 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|