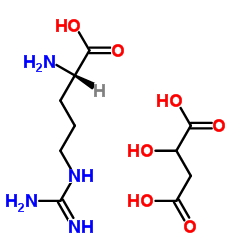
63-91-2
| Name | L-phenylalanine |
|---|---|
| Synonyms |
Benzenepropanethioic acid,S-phenyl ester
(S)-phenylalanine L-Phenylalanine EINECS 200-568-1 (2S)-2-amino-3-phenylpropanoic acid L-PHENYLALININE α-Amino-β-phenylpropionic acid, L- H-Phe-OH Alanine, 3-phenyl- L-phenylalanine zwitterion S-phenylalanine (S)-(-)-Phenylalanine Thiohydrozimtsaeurephenylester (S)-a-Amino-b-phenylpropionic Acid Alanine, phenyl-, L- L-2-Amino-3-phenylpropionic acid (S)-2-Amino-3-phenylpropionic acid (S)-2-Amino-3-phenylpropanoic acid C6H5CH2CH(NH2)COOH (S)-a-Aminohydrocinnamic Acid Phenylalanine α-Aminohydrocinnamic acid, L- (L)-Phenylalanine Phenylalanine (VAN) Phenylalanine, L- Benzenepropanoic acid, α-amino-, (S)- l-Phe PhE phenyl 4-phenylthiobutanoate S-Phenyl-3-phenylpropanthioat β-phenyl-L-alanine (-)-Phenylalanine (S)-α-Aminobenzenepropanoic acid MFCD00064227 3-Phenylpropionyl-phenyl-thioether α-Amino-β-phenylpropionic acid 3-Phenylthiopropionic acid,S-phenyl ester (S)-A-Aminobenzenepropanoic Acid Acetylcysteine Impurity 3 |
| Description | L-Phenylalanine is an antagonist at α2δ calcium channels with a Ki of 980 nM. IC50 Value: 980 nM [1]Target: Calcium ChannelL-Phenylalanine (LPA) is an electrically neutral amino acid, one of the twenty common amino acids used to biochemically form proteins. In the brain, L-phenylalanine is a competitive antagonist at the glycine binding site of NMDA receptor and at the glutamate binding site of AMPA receptor [2, 3]. At the glycine binding site of NMDA receptor L-phenylalanine has an apparent equilibrium dissociation constant (KB) of 573 ?M estimated by Schild regression [4] which is considerably lower than brain L-phenylalanine concentration observed in untreated human phenylketonuria [5]. L-Phenylalanine also inhibits neurotransmitter release at glutamatergic synapses in hippocampus and cortex with IC50 of 980 nM, a brain concentration seen in classical phenylketonuria, whereas D-phenylalanine has a significantly smaller effect [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 307.5±30.0 °C at 760 mmHg |
| Melting Point | 270-275ºC (dec.)(lit.) |
| Molecular Formula | C9H11NO2 |
| Molecular Weight | 165.189 |
| Flash Point | 139.8±24.6 °C |
| Exact Mass | 165.078979 |
| PSA | 63.32000 |
| LogP | 1.11 |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.576 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | C: Corrosive; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 22-24/25-37/39-45-36/37/39-27-26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | AY7535000 |
| HS Code | 29224995 |
| Precursor 6 | |
|---|---|
| DownStream 10 | |
| HS Code | 29224995 |
|---|



![3-phenyl-(2S)-[(1'R)-phenylethylamino]propionic acid structure](https://image.chemsrc.com/caspic/203/1004755-04-7.png)


![Benzenepropanoic acid,-[(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)oxy]-,(aR)- structure](https://image.chemsrc.com/caspic/117/310404-47-8.png)